A Maggot Revolution In Modern Medicine
11:14 minutes

 Do you want to go back in time with SciFri? Sign up for the Science Friday Rewind newsletter to get more never-before digitized stories and audio bites from our archives!
Do you want to go back in time with SciFri? Sign up for the Science Friday Rewind newsletter to get more never-before digitized stories and audio bites from our archives!
In a bloody battle during World War I, two wounded soldiers were stranded on the battlefield in France, hidden and overlooked under some brush. Suffering femur fractures and flesh wounds around their scrotum and abdomen, they lay abandoned without water, food, or shelter for a whole week. At the time, outcomes for these kinds of wounds were poor: Patients with compound femur fractures had a 75 to 80% mortality rate. By the time the soldiers were rescued and brought to a hospital base, orthopedic surgeon William Baer expected their wounds to be festering, and their conditions fatal. But much to his surprise, neither showed any signs of fever, septicaemia, or blood poisoning.
When his team removed the soldiers’ clothing, they discovered that their flesh wounds were filled with thousands of maggots, or baby flies—little larvae with a massive appetite for decaying matter. Baer was repulsed by the sight, and the team quickly washed off the wriggling maggots. Underneath, instead of the expected pus and bacteria-infected flesh, Baer marveled over “the most remarkable picture.”
“These wounds were filled with the most beautiful pink granulation tissue that one could imagine,” Baer later wrote in a 1931 report in the Journal of Bone and Joint Surgery. Maggots have long been associated with death, but in this case, they were helping the soldiers stay alive. As these insects were simply tucking in for their typical meal of dead, decaying flesh, they were inadvertently aiding the soldiers by cleaning their wounds, keeping infection at bay. The soldiers recovered—saved by their tiny, wriggling “friends which had been doing such noble work,” Baer wrote.
Baer’s paper is one of the first reports of maggots used in medicine, but these insects have been found healing wounds for thousands of years, with references in the Old Testament and in ancient cultures of New South Wales and Northern Myanmar. In 1997 on Science Friday, authors Robert and Michèle Root-Bernstein regaled the audience with tales of maggots and other historical medical practices:
“In the ’30s, it became a very useful and effective therapy,” Michele Root-Bernstein explained.
After the war, Baer brought his observations back to America, experimenting with applying maggots to surgical operations on chronic osteomyelitis, a debilitating bone disease that can persist for years. Some of his studies were more successful than others; in his early trials attempting to replicate what he saw on the battlefield, he collected unsterilized maggots from rotting raw beef he left outside, which led to some secondary bacterial infections. But once Baer established which species of fly worked best, and how to sterilize them, he observed that maggots could cure patients’ lesions and sores within months—fast compared to other available treatments, which could take a decade.
His experiments hatched a maggot revolution in modern medicine. Over 300 hospitals in the U.S. and Canada began using medical maggots in surgeries and wound treatment. The mass production and convenience of penicillin in the 1940s caused the maggot fad to fade over time, but in the 1990s, they made a “resurgence,” Root Bernstein said, in part because of the rise of antibiotic-resistance.
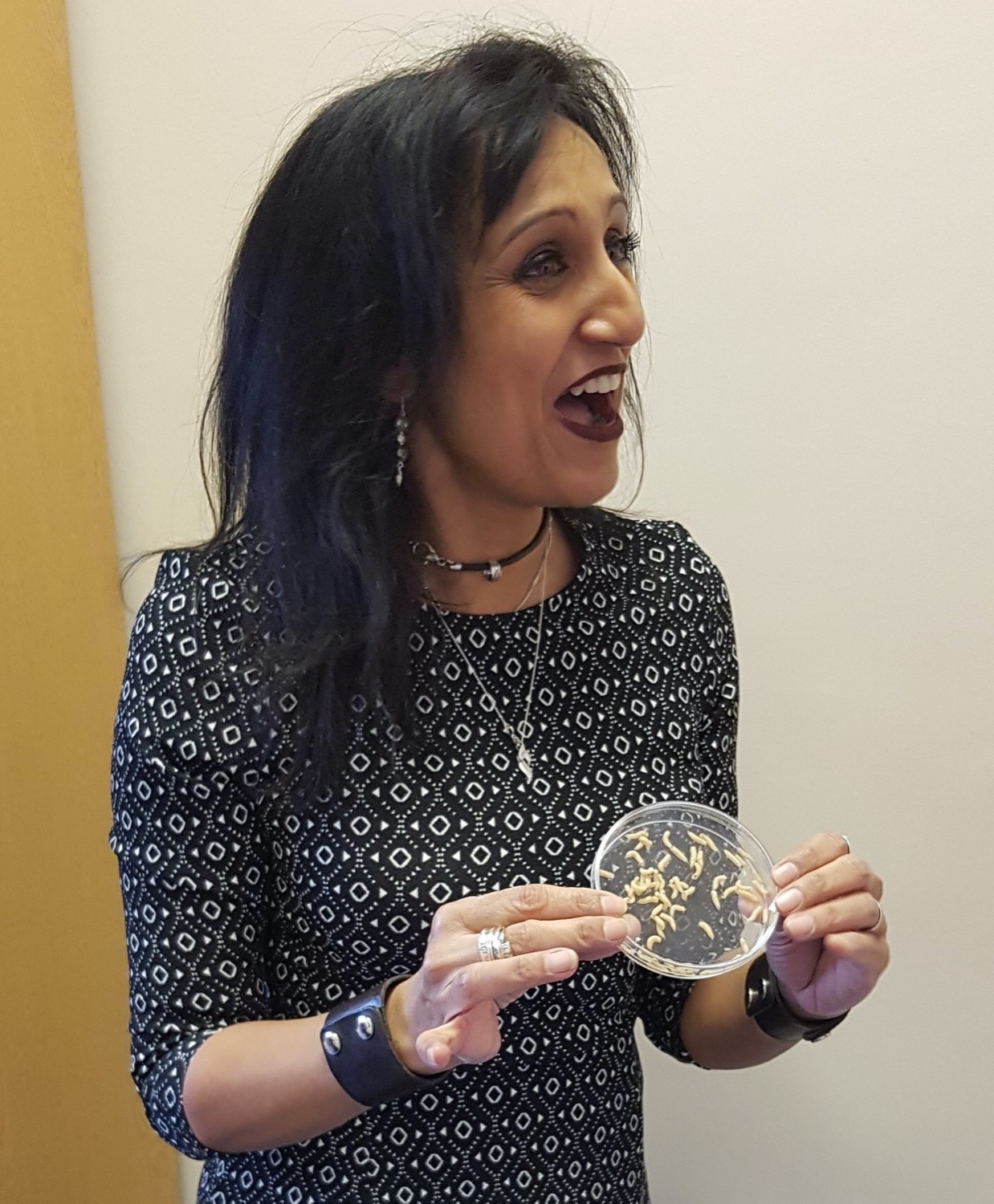
Today, medical maggot therapy is a clinically approved practice in countries around the world, including the United States, United Kingdom, China, Australia, and many European countries. Under medical supervision, maggots debride wounds, or remove damaged and infected tissue. They are still used to treat an array of chronic wounds, including diabetic foot ulcers, venous leg ulcers, gangrenous bed sores, and even burns. Some studies suggest that when the therapy is used as a last resort before amputation, maggots help save 40-50% of limbs—although it might be even more effective if used earlier in the course of treatment, says Yamni Nigam, biomedical science professor at Swansea University in the United Kingdom.
Now, clinicians and researchers like Nigam are studying the unique molecular cocktail that maggots secrete in order to better understand how they aid healing. “They are fantastic and very speedy and efficient wound cleansers and debriders,” Nigam tells Science Friday in a recent phone interview. As a medical entomologist who has been studying maggot therapy since 1997, she says their role is simple: “Any dead tissue, they will physically clear it away and destroy any infection that’s there.”

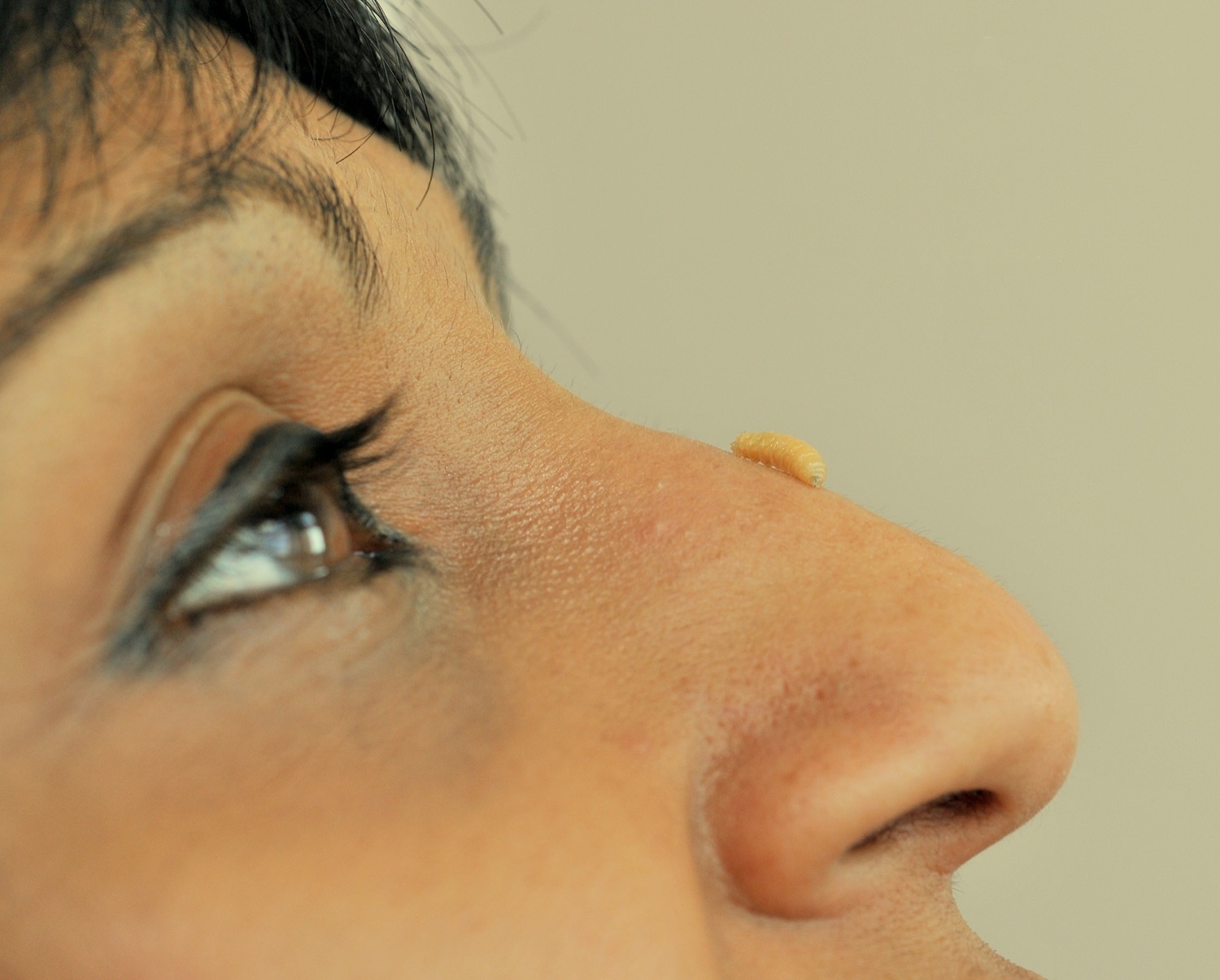
But the medical maggots Nigam studies are not the typical larvae you might find squirming in rotting roadkill or your spoiled garbage. Medical maggots are bred in special conditions, often from the shimmery common green-bottle fly, also known as Lucilia sericata. Other species can eat healthy tissue and invade the rest of the body, but Lucilia sericata only target dead flesh, making them prime candidates for clearing and debriding wounds. In an aseptic environment, factories breed, sterilize, and feed the maggots a very strict diet. When freshly hatched, the baby maggots are only a few millimeters long—smaller than a grain of rice.
“The maggots produced are really brilliant, clean little things,” says Nigam. Medical maggots do not bite, chew, or have any teeth. Instead, “they secrete enzymes, like little chemical scissors, which break down the dead tissue into a soup,” Nigam says. “Then they just drink [up].”


The maggots are transported to hospitals when they are only one to two days old, when they are at their hungriest. If a patient’s case and condition is a good fit, up to a several hundred of the insatiable insects are applied directly to wounds with a dressing system that keeps them in place, or sealed into a so-called BioBag, a woven pouch with a piece of foam that sits atop the wound, where the maggots’ secretions seep on to the dead tissue. While there are cheaper (and more expensive) therapies, Nigam explains studies have shown that the insects’ efficiency outweighs their cost.
“What those maggots can do within three days is what sometimes takes years of time to achieve with other therapies,” says Nigam, explaining that medical maggots can clear an infection in as little as two to three days.

The maggots are often left on wounds for four days, after which they are typically removed and incinerated—long before they transition into their adult stage, as the green-bottle fly larva takes about two weeks to transform into flies. Full treatment can take from one to 12 sessions, depending on the extent of the wound.
“They secrete enzymes, like little chemical scissors, which break down the dead tissue into a soup.”
The exact biochemical activity involved in maggots’ wound-healing abilities has remained a mystery until recently. In his 1931 report, Baer mused that the maggots were creating some “biological reaction” that played an important role in healing, and guessed that it involved a “biochemical substance.” The answer, Nigam now says, lies in the maggots’ spit and secretions.

In 2003, Nigam began breeding her own colony of maggots in the lab to find out the molecular makeup of these secretions. Her team set up cages of green-bottle flies, gathered the eggs, and placed hatchlings into test tubes, where they washed them to collect their sweat, saliva, and bodily secretions. Then the researchers ran tests to find out if there were molecules in these fluids that could combat infectious bacteria strains.
“We discovered that maggot secretions did have very brilliant and potent antibacterial activity,” Nigam says.
One of the molecules, that the team named Seraticin, is effective at killing bacterial strains of difficult-to-treat infections, including methicillin-resistant S. aureus (MRSA) and E. coli, which now have high resistance to antibiotics. This secretion is also packed with molecules that trigger fibroblasts—which promote collagen and elastin—to travel faster across a wound bed, speeding up the healing process. The team also found the fluid had antifungal properties, and a couple years ago, another research group discovered activity against certain viruses, such as the Rift Valley Fever virus.
“If you put a maggot on the wound, it’s [like] a factory,” she says. “You’re giving the patient everything.”
This suite of antifungal, antiviral, and antimicrobial properties makes evolutionary sense, says Nigam. “Maggots have been living on this Earth for millions of years, and they’ve had to evolve with the organisms that are also feeding on dead corpses,” she says. In this feast of necrotic flesh, maggots must protect themselves from the other decomposers dining at the table. “The maggots have developed their own systems and their own molecules to fight bacteria and fungi,” she says. “All we’ve done is exploit that.”
A maggot’s ability to adapt to threats also makes them incredibly useful when it comes to antibiotic-resistant bacterial strains, when germs develop the ability to defeat the drugs designed to kill them. Some researchers have considered isolating the molecules from the secretions to create new antibiotics, but Nigam says those medicines could also eventually become ineffective—unlike living insects. “This is the beauty about having a living creature sitting on you,” Nigam says. “The chances are, it’s evolved to deal with any bacteria and fungi in a wound that have mutated.”

In recent years, Nigam has focused on dispelling myths about maggot therapy, educating the public about its safety and effectiveness. Despite their benefits and availability, medical maggot therapy still faces a big hurdle: Public fear and disgust. The treatment isn’t as common as it could be, due to the reluctance of patients, nurses, and doctors.
People often ask Nigam if the maggots will eat them alive, or turn into flies during the procedure (which Nigam reiterates, doesn’t happen). In a 2017 survey, Nigam even found that some people would rather undergo amputation than try maggot therapy. To help change maggots’ negative reputation and get more patients to consider the therapy as an option, Nigam launched the Love A Maggot! campaign in 2016. She conducts studies to understand people’s attitudes towards maggot therapy, and speaks with medical students, nurses, clinicians, patients, young children, and the general public.
At her events, she brings models, props, and, of course, “I always have living maggots with me,” she says. She’ll let the larvae crawl in people’s hands. “They’re really quite cute,” Nigam says, adding that once they understand what maggots can do, she thinks “people [will] start loving them like I do.”
Archival interview excerpts have been edited for length.
Invest in quality science journalism by making a donation to Science Friday.
Yamni Nigam is a lecturer in Biomedical Science at Swansea University in Swansea, UK.
Lauren J. Young was Science Friday’s digital producer. When she’s not shelving books as a library assistant, she’s adding to her impressive Pez dispenser collection.
SOPHIE BUSHWICK: When a baby fly hatches, it has one job, and that job is to get as big as possible as fast as possible. This is why we often find those babies– all right, they’re maggots– in organic matter like dead animals or sometimes our trash. Hey, a kid’s got to eat. That voracious hunger and taste for dead flesh is one reason maggots have been used to help heal wounds since antiquity. It turns out they work really, really well at getting infections out of the way so the wound can begin to close.
But although maggots went out of fashion shortly after the invention of antibiotics, researchers want you to know that they’re an old school remedy with increasingly appreciated benefits in the era of antibiotic resistance. Here with more is SciFri digital producer and archive dweller, Lauren Young. She’s the mind behind a new piece up on the SciFri website about the recent and ongoing advances in medical maggots. You can check that out on our website, sciencefriday.com/maggots. Hi, Lauren.
LAUREN YOUNG: Hey, Sophie.
SOPHIE BUSHWICK: What got you looking into the story of medical maggots?
LAUREN YOUNG: All right, yeah, so it was a dark and stormy night. No, just kidding. I was poring through the SciFri archives, digging around for stories for our series SciFri Rewind, and I stumbled upon this in a 1997 conversation Ira had with author Michelle Bernstein.
IRA FLATOW: When most people think about maggots, they probably think about something that people used before they knew better. But I was surprised to learn and interested to learn that doctors are actually still using maggots to do something, right?
MICHELLE BERNSTEIN: They are. Maggots have been worming their way back into clinical practice.
LAUREN YOUNG: So thanks to this intriguing tidbit, medical maggots sort of wriggled their way into my curiosity. So I really wanted to find out more what researchers have discovered since this 1997 conversation. I had reached out to Dr. Yamni Nigam, a biomedical researcher and lecturer at Swansea University in the UK. She’s been studying these tiny fly larvae since the late 1990s, and she’s a big fan.
YAMNI NIGAM: Most people are like, oh, wow. Oh, they feel really amazing, and they’re really cute. Which is something that I’ve always said about maggots.
SOPHIE BUSHWICK: I definitely never thought of them as cute before. But when you watch them wiggle around long enough, they certainly grow on you. Not literally, of course.
LAUREN YOUNG: Right? Yeah, and more importantly, maybe they can actually help us. So when maggots feed on dead flesh and decay, they have to eat alongside other decomposers like bacteria and fungi. So it’s caused them to evolve some cool protective chemicals that also happen to benefit us. Yamni told me about the research of William Baer, a doctor who treated soldiers during World War I. He observed that soldiers who had maggots in their wounds were remarkably free of infection, even if they had gone days without medical care. It was–
SOPHIE BUSHWICK: Oh, wow.
LAUREN YOUNG: Yeah, super fascinating observation.
SOPHIE BUSHWICK: That’s really interesting.
LAUREN YOUNG: Yeah. So the thing Yamni and other researchers are learning now, though, is why maggots are so good at healing wounds.
SOPHIE BUSHWICK: And Yamni was so great to talk to once that we had to call her up again. I interviewed her earlier this week. We started out by talking about how maggots can make a difference in the wound healing process.
YAMNI NIGAM: Maggots are very speedy debriders. They are nature’s debriders. And by debridement, we mean getting rid of dead necrotic tissue. If a wound has dead necrotic tissue, debris of old skin and so on, it won’t heal. It will never progress. If a wound is infected, it will never progress through the stages of healing. What maggots do very, very effectively is they remove the necrotic tissue, and they absolutely get rid of the biological burden, the bacteria in the wound, and they kickstart the healing process. So they have plenty of roles to play within wound healing and wound debridement.
SOPHIE BUSHWICK: Yamni, I mentioned earlier that we stopped using maggots when penicillin was invented. But now they’re hot again, aren’t they?
YAMNI NIGAM: Indeed they are. I think the fact that we have so many resistant strains of bacteria that are not responding to our antibiotics anymore, and they’ve evolved methods and ways of evading our antibiotics. And yet, if you put maggots in a wound that has a resistant infection, that infection will be cleared up. So people are beginning to look back to maggots because they know that they actually can treat resistant infections in wounds.
SOPHIE BUSHWICK: And your work in particular is looking at why that is. So what have you learned about that?
YAMNI NIGAM: So we’ve been looking at a couple of things. Our main focus at Swansea has been looking at how exactly are maggots clearing a wound infection. We know that they can do it, but we didn’t know how. And it’s only recently that we’ve discovered that maggots actually secrete in their spit and sweat, if you like. It’s a excretion secretion, really. They actually produce these antibacterial molecules. And there are vast numbers of these molecules. Lots of them are actually tailored to the wound infection.
So if you put a maggot in a wound that has a particular species of bacteria in it, that maggot will up its game to produce molecules that will specifically destroy that particular infection. That’s called the inducible maggot activity. And many researchers across the world have shown this. But we, indeed, in Swansea, have identified a particular small molecule that we’ve trademarked that’s called seraticin that we know killed MRSA and kills lots of other different types of bacteria that are present within the wound.
SOPHIE BUSHWICK: And has there been any movement to take some of those compounds that you know they secrete and just use that directly, instead of just putting the maggots on the wound?
YAMNI NIGAM: I think you have to watch this space, really. Certainly, that’s a huge goal of scientists, clinicians. The public, I think, in general, would prefer to have a secretion-based ointment, let’s say, rather than the real life maggots. But I myself have to say that I think the real thing is a factory of molecule production. It’s producing whatever it needs in that wound. Not only is it producing enzymes that will digest the dead tissue, it’s producing antibacterial molecules. But we also know maggots produce molecules that aid healing.
So if you’ve put the whole package together, you’ve got a factory, a maggot factory on your wound. For three to four days, that’s as long as we leave them. And you’ve got very good beneficial effects from the whole thing.
SOPHIE BUSHWICK: Just a quick reminder that I’m Sophie Bushwick, and this is Science Friday from WNYC Studios, talking to biomedical researcher Yamni Nigam about the medical magic of maggots. And what would you tell someone who could benefit from maggot therapy, but is maybe a little reluctant to try it? What would you tell them about what the process is like and what it feels like to have this therapy?
YAMNI NIGAM: The process is very simple in the sense that if the wound is suitable for maggot therapy and the clinician will assess that, they will then put a tiny little baby, the cutest little baby maggots. They’re a millimeter. And they will go on the wound usually in small polystyrene bags. And that’s sealed so the maggots don’t get out.
And these enzymes come out of the bag. They go out onto the wound to the dead tissue. They turn the dead tissue into a digestible soup almost for themselves, and then they drink that up. And that happens within two to three days very, very quickly. So that’s a process. The bag is then removed after three or four days. And the maggots are then removed from the patient. And the wound usually is absolutely sparkling clean at that point. So it is a very quick, efficient, and very effective process.
The feeling varies between patients. Some patients don’t feel a thing, and some patients say it tickles. And then some patients feel pain. And often, we find that patients that feel pain might have some underlying pathophysiology, or they might be very reluctant to have used maggots. And therefore, they have a negative association anyway. So we’re finding all sorts of things. We are investigating, really, how people react to maggots.
SOPHIE BUSHWICK: And if stigma is part of what’s holding back research and the use of maggots in medicine, what do you think those who think that maggots are cute need to do to warm more people up to that point of view?
YAMNI NIGAM: We’ve launched what we call a love of maggots campaign. We’ve got websites of it. And I go out to the public. I go out to not just the general patients, but I go out to nurses and doctors, too, because they often are also reluctant to use it. They’re a little bit squeamish as well. And so I think it all depends on changing a mindset. It’s increasing awareness that maggots can work really, really effectively, increasing acceptance, changing the negative perception.
And one of the ways that we’ve tackled this is by going into schools. So if you put a little maggot on a three-year-old’s hand, they’ll be like, oh, that’s so cute. But if you put a maggot on a nine-year-old’s hand, they’ll be like, ew, get that off me. So somewhere along the line comes an association with negativity, whether it’s parents, whether it’s the child themselves associating maggots with smell in the dustbin or dirt or whatever. And so we need to go into primary schools.
And really, we need to show children how brilliant this particular medicinal maggot is and how useful it can be, so that when the child grows up or when the child goes home, and his grandparents have chronic wounds and leg ulcers and diabetic ulcers, he can actually say, but yes, I’ve learned that these maggots are brilliant and they’re very beneficial. So we are tackling all aspects, all sides, really, to try and get it more accepted by the public.
SOPHIE BUSHWICK: Is part of this stigma connected with the name maggot? And do you think that maybe there’s a different name that we could be using? We’ve had this chat repeatedly with a lot of people. One of my teams said, why don’t we call them high genies or something, a different name? And that’s brilliant, but when you say, right, we’re going to put high genies on you, and then the patient says, well, what’s that, then you have to say, well, they’re maggots.
So I don’t think you can get around it. I think that the stigma is– you’re right, the name maggot does instill fear and repulsion in a lot of people. But I think they need to be aware that this species is a good species of maggot. This will really, really help wounds to debride, to disinfect, to heal. So I think it’s all about explaining to patients, really, rather than just trying to disguise it, I think.
SOPHIE BUSHWICK: And I’m afraid we have to leave it there. Thank you so much for joining us.
YAMNI NIGAM: You’re very welcome. A pleasure. Thank you very much, indeed.
SOPHIE BUSHWICK: Dr. Yamni Nigam, a lecturer in biomedical science and self-described professor of maggots at Swansea University in the UK. You can learn more about her research and the status of maggots in medicine by checking out our producer Lauren Young’s excellent piece on our website, sciencefriday.com/maggots. Thanks for that story, Lauren. This is all so cool.
LAUREN YOUNG: You’re welcome, Sophie. I’m never going to look at a maggot the same way ever again.
Copyright © 2021 Science Friday Initiative. All rights reserved. Science Friday transcripts are produced on a tight deadline by 3Play Media. Fidelity to the original aired/published audio or video file might vary, and text might be updated or amended in the future. For the authoritative record of Science Friday’s programming, please visit the original aired/published recording. For terms of use and more information, visit our policies pages at http://www.sciencefriday.com/about/policies/
Lauren J. Young was Science Friday’s digital producer. When she’s not shelving books as a library assistant, she’s adding to her impressive Pez dispenser collection.
Christie Taylor was a producer for Science Friday. Her days involved diligent research, too many phone calls for an introvert, and asking scientists if they have any audio of that narwhal heartbeat.
Sophie Bushwick is a freelance science journalist and editor based in New York.