Pitting Viruses Against Bacteria To Combat The Antibiotic Crisis
17:19 minutes
[Scientists discovered a way to create a rare natural phenomenon in the lab: ball lightning.]
This week in a study in Evolution Medicine and Public Health, biologist Benjamin Chan and his team describe using phage therapy to treat a patient who developed an antibiotic-resistant infection. Chan and bioengineer Timothy Lu talk about developing both natural and synthetic phage treatments and how these drugs could be used to combat antibiotic resistance
Benjamin Chan is an associate research scientist in Ecology and Evolutionary Biology at Yale University in New Haven, Connecticut.
Timothy Lu is an associate professor of Electrical and Biological Engineering at Massachusetts Institute of Technology in Cambridge, Massachusetts.
IRA FLATOW: This is Science Friday. I’m Ira Flatow. Our next story is about a virus fished out of a lake that healed a man, suffering from an antibiotic-resistant infection in his heart– saved his life. A little backstory first. You’ve heard that there’s an antibiotic crisis, right?
We all know that. More and more bacteria are becoming resistant to the standard drugs. In fact, in the US alone, two million people each year develop infections that cannot be treated by the usual antibiotics. So the rush is on to find new antibiotics.
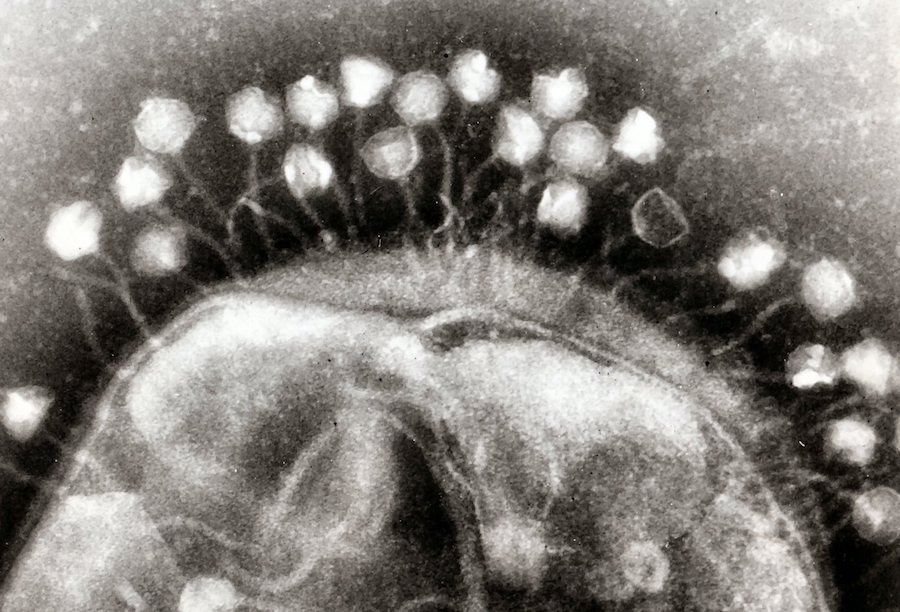
Some scientists, though, are turning to an old technique that was being investigated before World War II, before the age of antibiotic wonder drugs, enlisting viruses called bacteriophages– viruses that seek out and kill bacteria in nature. They do it all the time.
Well, now back to that patient. He was given a dose of a bacteriophage, a virus found in a lake, and he healed. He was healed in a matter of days. The scientists who found that phage published their results this week in the journal, Evolution, Medicine, and Public Health. So, could we engineer phages for novel treatments?
Why not let nature do the work for us? And, are we finally in the age where we can say the phage is all the rage? I had to get that in there. Sorry. Let me introduce my guest. Benjamin Chan is an author in that study from Yale University. Welcome to Science Friday. Hi, Benjamin. Are you there?
BENJAMIN CHAN: Yeah, I’m here.
IRA FLATOW: Hi.
BENJAMIN CHAN: Thanks for having me.
IRA FLATOW: Well, I also bring on Timothy Lu, associate professor of electrical and bioelectronic engineering at MIT. Welcome to Science Friday.
TIMOTHY LU: Hi, there. Thanks for having me.
IRA FLATOW: Hi, there. Now, Benjamin, this patient you treated came to you as a last resort. How did you hear about this patient?
BENJAMIN CHAN: I heard about him through sort of the hospital administration. When I first popped into the university, I sent emails all over and saying, hey, I’m new here. I’m really interested in doing some of this stuff.
And the former head of the hospital got back to me and said, I think you should talk to these people– forwarded me along and to Deepak Narayan, also an author on the study. When I spoke to him in his office, he said, I’ve got a case you could– you should look at and see if we can do something about this.
IRA FLATOW: Did you already know that you had a virus, a phage that could combat this infection?
BENJAMIN CHAN: Well, I had lots of bacteriophages, right?
IRA FLATOW: Yeah.
BENJAMIN CHAN: So when I first arrived, I started collecting them from all over different sources. But I didn’t know for sure that it could work on his strain, until I actually got the strain from him.
IRA FLATOW: So, you injected the phages directly into the infected area, right?
BENJAMIN CHAN: Close.
IRA FLATOW: Well, how did you know how to do it, and where to put it? What size dosage? All those things.
BENJAMIN CHAN: Well, we didn’t, to be totally honest, because it’s not known. It hasn’t been done before in this sort of indication. And so we were sort of– it was a lot of guesswork. But that was based on a lot of evidence that we sort of came up with in the lab, where we tested the minimum number of phages required to kill an approximate number of bacteria which we estimated. And so we did a lot of prework in the lab and then just estimated that maybe 10 to the eighth would be sufficient.
IRA FLATOW: And now, you’re a phage hunter, right? You go out into the slime and the muck, and you look for these naturally-occurring viruses?
BENJAMIN CHAN: Yeah, I go all over. A lot of times it’s the muck and the slime, but sometimes it’s nice lakes and rivers as well.
IRA FLATOW: And this one was found in a lake?
BENJAMIN CHAN: Yeah, in a lake here in Connecticut.
IRA FLATOW: Wow. Dr. Lu, you’re a synthetic biologist. You’re engineering phages to get around the antibiotic-resistant problem. How do you build these phages? Do you use the phage code, the package that you want that’s inside there? How do you make a phage?
TIMOTHY LU: Well, what we do is take the genetic material that’s inside that phage. It basically is the software program that actually encodes all of those phage components. And our focus is, then, how do you pluck things out of that phage genome for your application of interest? Or, how do you insert new materials into that phage genome, so that you can endow that phage with new functionalities? For example, use in therapeutics or even as diagnostics.
IRA FLATOW: Did you need Benjamin’s library to be able to build your phages?
TIMOTHY LU: Well, I think, certainly, Benjamin’s library, and many of the libraries that are being discovered by the phage hunters, are really valuable for this sort of work that synthetic biologists want to do. There’s amazing diversity of genetic material out there that have evolved over time. And these phages have been countering bacteria and vise versa. And that starts as a great starting substrate for the types of engineering that we’d like to be able to do.
IRA FLATOW: Our number– 844-724-8255. You can also tweet us @scifri, talking about using phages. As I mentioned before, this is not a new idea. Didn’t the Soviets work on this before World War II and have laboratories where they were trying to create bacteriophages that were– sort of went out of fashion when the areas of these miracle antibiotics came about?
BENJAMIN CHAN: Sure. Yeah. Bacteriophages have been developed as therapeutics for almost 100 years now. And the work persisted in the former Soviet Union, where it faded out here, after the discovery of broad-spectrum antibiotics.
IRA FLATOW: And when you go out to look for phage, Benjamin, do you go looking for a specific thing, or you just go out there and say, hey, let’s see what I can pull out of the lake today?
BENJAMIN CHAN: A little bit of both, I guess. For some of the phages we’re looking for, we have a pretty good idea where they won’t be, and so we can sort of tailor our search. We study cholera as well. And so you have to go to a location that actually has cholera in order to find the phages.
IRA FLATOW: And, Tim, what do you do to create a new phage? Give us a process that’s involved.
TIMOTHY LU: Yeah. So as an example of some work that we’ve done before, is to take that phage genome, which nowadays with DNA sequencing technology has been fully read. And so we can then identify specific locations where we’d like to pop in additional genes that would endow, for example, that phage with the ability to break up biofilms more effectively, or reduce that bacteria’s ability to develop antibiotic resistance.
Then depending on what tools you want to use, we can use tools like CRISPR to cut out DNA from that phage and insert our new DNA in that, or even if we can use some of the new DNA synthesis-based technologies and print that DNA from scratch, and actually insert that into the phage genome.
We then take that now recombinant, or a new phaged genome, and we can then insert it back into the bacteria that actually is the host for that phage. And when that DNA goes in, it sort of boots itself up and can generate a new copy of that bacteriophage, now with the additional functionality that we’ve encoded into that bacteriophage.
IRA FLATOW: I find it ironic that you use CRISPR against the bacteria, because CRISPR is what the bacteria uses against the virus, isn’t it?
TIMOTHY LU: Yeah. I think the amazing thing about phage biology in general is not only has it this great potential as a therapeutic agent, but it’s actually phage biology. Plus, the interaction between phages and bacteria has really been a really rich source of all the tools that we actually use in modern-day molecular biology, ranging from some of the things that gave rise to CRISPR, or those studies that gave rise to– of our basic understanding of molecular biology and how to manipulate it. So these phages are everywhere in the environment. And I think they’ve really made a big impact in biology, even beyond the use in antibiotic resistance.
IRA FLATOW: Benjamin, so, how was this particular phage you used– how was it able to knock out this type of bacteria?
BENJAMIN CHAN: So this particular one we used as sort of a way to drive– force a trade-off between antibiotic sensitivity and phage sensitivity. Because early on, we found that when bacteria became resistant to this phage, that they suddenly were now susceptible to antibiotics. And so we forced sort of a trade-off, where we anticipated bacteria becoming resistant to this phage. But as a consequence, it would be susceptible to antibiotics.
IRA FLATOW: Why aren’t more drug companies doing this kind of thing?
BENJAMIN CHAN: Well, I mean, it’s complicated, I guess, like all things. But I think a major part of the problem, I guess for a company whose interest is profit, is sort of intellectual property. As far as I can tell, that’s a bit harder when you’re looking at live organisms. But I think with synthetic organisms or whatever– engineered ones– you can get around that problem.
IRA FLATOW: Tim, can you patent a phage that you’re making?
TIMOTHY LU: Yeah, I think you can get intellectual property coverage over things that are manipulated, so I think that is one piece of the puzzle. But as Benjamin also mentioned, it’s a complicated problem. I think, in general, antimicrobial drug discovery is underfunded compared to other areas of research.
And if an antibiotic works the way it should, it should cure that patient in just a few days. And it’s not something that’s going to generate years or decades of profits for pharmaceutical companies. So I think in terms of the way the current incentive system is set up, it’s not really conducive for the types of profit-focused drug development that many companies focus on.
And so there have been interesting discussions about alternative approaches to try to incentivize antimicrobial drug development, since it’s really– if you think about antimicrobials– are sort of a public good, in that if I use an antibiotic too much, it actually affects your future ability to use that same antibiotic. And so maybe we have to think about more creative ways to incentivize drug development in this area.
IRA FLATOW: Is one of the obstacles that these phages are very specific to one bacterium? These aren’t broad-spectrum treatments? You have to make it– it’s not a shotgun? It’s a very precise shot here, Benjamin?
BENJAMIN CHAN: I mean, it can be. I think that’s a massive benefit, because you’re not going to disrupt any of the microbiome. And you can use the specificity of bacteriophages to sort of target specific proteins, or sugars, or whatever you’re looking at– or virulence factors on the bacteria surface, so that when they become resistant, you’ve sort of made them less able to cause disease or antibiotic susceptible. So I like– I think the specificity is a massive benefit for using phages therapeutically.
IRA FLATOW: We talk a lot about the microbiome here, the bacteria that’s in our gut a lot. I would imagine– tell me if I’m wrong. If there are all these bacteria in there, there got to be phage viruses trying to attack them, also. So we have a lot of viruses lurking around in our gut. Can we engineer phages that might help our microbiome? Tim, what do you think?
TIMOTHY LU: Yes. I think, certainly, there are a lot of phages and other viruses that are actually present in our microbiome. They’re relatively understudied compared to the bacterial members of that community. But if you think about it, many of the approaches we have now to manipulate the microbiome, including antibiotics, are not very narrow spectrum.
There’s no really ready way for us to say, I want to knock out this one specific bug from inside my gut. And so I think the development of narrow spectrum, targeting it just like bacteriophages, can be very useful in that process to really allow us a greater toolset, to be able to manipulate the bacteria that are inside of us.
IRA FLATOW: Let’s go to the phones– 844-724-8255 to South Bend, Indiana. Hi, Brian.
BRIAN: Hey, I’m curious. Is there any chance for these phages becoming pathogenic to the patient? And if so, how do you manage that immune response that the patient would have against the treatment?
IRA FLATOW: Good question.
BENJAMIN CHAN: So, I mean, as far as we know, there’s no bacteriophages that can infect or attach to human cells, since they’re very, very, very specific to particular strains, often of bacteria. And managing the immune response– maybe Dr. Lu could answer, since he’s a physician.
TIMOTHY LU: Yeah. I mean, I think the immune response for foreign elements in the body is certainly something to consider. I think in many of these cases, giving them as an acute treatment– as we’ve seen from some of the studies that Benjamin has published and others, I think there’s an immediate, sort of risk-benefit to be able to use these, especially when there’s infections that are not present.
There have been strategies to try to shield these bacteriophages from the immune system, covering them with certain polymers, or actually mutating the phages, so that they can hide from the immune system to a greater extent.
To my understanding, there haven’t been significant adverse effects– been reported with the administration of phages from that perspective. But it is a concern that I think anyone who is using these should be aware of and continue to monitor.
IRA FLATOW: I’m Ira Flatow. This is Science Friday from PRI, Public Radio International, talking with Benjamin Chan and Tim Lu about the phages. What if a doctor has a patient and wants to get in contact with you, Benjamin, and say, here’s the story, and says, I have patients all the time who are at the end, who have run out of antibiotics? Are you open to hearing from them?
BENJAMIN CHAN: Sure.
IRA FLATOW: And, is there a databank? Is there a place for people to go?
BENJAMIN CHAN: Yeah. Of course, I’m open to hearing from them. I actually heard from one late last year. I mean, I hear from them fairly regularly, but we actually treated a second case in December of this year with some great results. So, I mean, google me– or I should be in the first 10 pages or so and email me.
IRA FLATOW: And I’m just surprised that we don’t hear more cases– one, two. With so many people, there’s so many infections that can’t be treated. I would think people would be flooded with calls looking for phage therapy. Does that surprise you that you’re not?
BENJAMIN CHAN: Yeah. I mean, I get quite a few calls for it, so I wouldn’t say– it’s not flooding my inbox. But I think it’s more of a process than we realize. I mean, the FDA is great, actually. The process works in getting approval for these.
But a lot of times it’s getting the strain from the person and sending it to a lab, where they’re going to find– test their phage on it. And there’s a little bit of a delay there, but that’s going to change in the future as it becomes more accepted and there are banks sitting around, or the phages are more characterized.
IRA FLATOW: Dr. Lu, am I just being naive about all of this?
TIMOTHY LU: Well, no. I think it is an area that is quite exciting. I think as Benjamin mentioned, there is a process currently in terms of the way it’s done. And I think there’s a lot more work to try to make it systematic and expand the scope of what can be achieved.
I think one of the current challenges is, as was alluded to earlier, really the diagnostic challenge. How do you know you have a phage on the shelf that will go after the bacteria of interest? And inherently, there’s a time lag associated with that.
And if we want to push the use of these phages to the front line of medicine, we need better tools to diagnose the infections rapidly, and then be able to pull the phages off a shelf, to know that those will be actually effective in the patients we want.
So I think over time, if we want to see the broad utility of this, despite the great work that the labwork Benjamin and others are doing, they won’t be able to service, I think, everybody in the country who might want to do this, especially once more people start hearing about this. And I think we’re going to need sort of more high throughput and quicker processes to be able to execute on the vision here.
IRA FLATOW: In the minute I have left, I have somebody on the phone like that– RB From Fort Dodge, Iowa. Hi, RB. Welcome.
RB: Good afternoon, gentlemen. I recently went to my doctor of infectious diseases. And he told me that I had MRSA, and I was probably going to be on doxycycline for a year. My wife who is in medicine shook her head at me, like this is not great news. I was surprised at how many people have MRSA. I didn’t even know what it was.
IRA FLATOW: I need a question quickly, because we’re running out of time.
RB: OK. Any thoughts they have about MRSA and phages?
IRA FLATOW: OK, why not use phages to treat MRSA? Go ahead.
BENJAMIN CHAN: I mean, it depends on the infection, of course, and the nature of it. But there’s lots of phages that can infect and kill Staph aureus, including MRSA. And it’s just sort of matching the right phage with the right infection.
IRA FLATOW: So it’s done on a case-by-case basis? That’s how you work?
BENJAMIN CHAN: Currently. I mean, if we get through trials, eventually it could be like an approved thing that you just sort of– would be available like Dr. Lu was mentioning. But right now, it’s case by case.
IRA FLATOW: Can we speed it up, Dr. Lu?
TIMOTHY LU: Well, I think if we have more funding for great support and really can incentivize this area, I think the technology is there. It’s just a matter of getting the regulatory– getting the manufacturing, getting the scientists together, and really coming up with a strategy for rolling this out.
IRA FLATOW: Great way to end this conversation. Benjamin Chan, associate research scientist in the Department of Ecology and Evolutionary Biology at Yale. Timothy Lu, associate professor of electrical and biological engineering at MIT. Thank you both for taking time to be with us today.
Copyright © 2018 Science Friday Initiative. All rights reserved. Science Friday transcripts are produced on a tight deadline by 3Play Media. Fidelity to the original aired/published audio or video file might vary, and text might be updated or amended in the future. For the authoritative record of Science Friday’s programming, please visit the original aired/published recording. For terms of use and more information, visit our policies pages at http://www.sciencefriday.com/about/policies/
Alexa Lim was a senior producer for Science Friday. Her favorite stories involve space, sound, and strange animal discoveries.