Grade Level
6 - 8
minutes
15 min - 1 hr
subject
Physical Science
Activity Type:
data collection, Engineering design challenge
This lesson and activity is part of the Chemical Educational Foundation’s® award-winning You Be The Chemist® Activity Guides, a free resource for educators and parents who want to introduce students in grades K-8 to chemistry and science concepts in a fun, hands-on manner. Learn more at www.chemed.org
Relevant standards, formulas and equations
View the full activity guide and safety guide

Rocketry has existed for hundreds of years. Although the technology has greatly improved and there are numerous methods for propelling a rocket, the simple science behind rockets has always been the same. To propel a rocket, some kind of force must push it forward. A force is the amount of push or pull on an object. The mechanical force that pushes a rocket or aircraft through the air is known as thrust. In this experiment, you’ll make a balloon rocket that is propelled by pressure. Pressure is the amount of force exerted on an area.
When you blow up the balloon, you are filling it with gas particles. The gas particles move freely within the balloon and may collide with one another, exerting pressure on the inside of the balloon. As more gas is added to the balloon, the number of gas particles in the balloon increases, as well as the number of collisions. While the force of a single gas particle collision is too small to notice, the total force created by all of the gas particle collisions within the balloon is significant.
As the number of collisions within the balloon increases, so does the pressure within the balloon. In addition, the pressure of the gas inside the balloon becomes greater than the air pressure outside of the balloon. When you release the opening of the balloon, gas quickly escapes to equalize the pressure inside with the air pressure outside of the balloon. The escaping air exerts a force on the balloon itself. The balloon pushes back in a manner described by Newton’s Third Law of Motion. That opposing force—called thrust, in this case—propels the rocket forward.

Materials
- Balloons
- Straws
- 10 feet of string
- Permanent marker
- Cargo (paper clips, bottle caps, candy, etc.)
- Cereal boxes, construction paper, or any other material to make lightweight cargo containers
- Tape, glue, scissors, and any other materials needed for construction
- Review the information in the Safety First section of the You Be The Chemist® resource guide
Experimental Procedure
As the students perform the experiment, challenge them to identify the independent, dependent, and controlled variables, as well as whether there is a control setup for the experiment. (Hint: As the amount of gas in the balloon changes, does the distance the rocket travels change?)
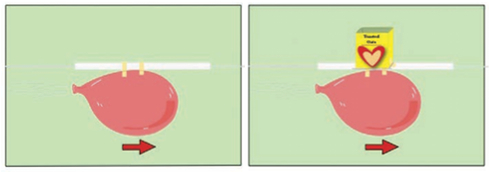
- Tie one end of a string to a chair, doorknob, or other support.
- Put the other end of the string through a straw. Then pull the string tight, and tie it to another support in the room.
- Blow up the balloon, and pinch the end of the balloon to keep the air inside. Do not tie the balloon.
- Tape the balloon to the straw so that the opening of the balloon is horizontal with the ground.
You may need two students for this: one to keep the air pinched inside the balloon and the other to tape the balloon to the straw. - While holding the balloon opening closed, one student should pull the balloon all the way back to the end of the string (the starting line) so that the balloon opening is against one support. Have another student use the marker to draw a finish line near the other end of the string.
- Let go of the balloon and watch it move along the string.
- Then, have students test different methods to transport “cargo” across the string to the finish line.
Data Collection
Have students record data in their science notebooks. What happened when the opening of the balloon was released and the gas was allowed to escape? If they timed the process, how long did it take for a rocket to cross the finish line? Have students answer the questions on the activity sheet (or similar ones of your own) to guide the process.
This lesson and activity is part of the Chemical Educational Foundation’s® (CEF) award-winning You Be The Chemist®Activity Guides, a free resource for educators and parents who want to introduce students to chemistry and science concepts in a fun, hands-on manner. The guides feature nearly 1,000 pages of lesson plans, science content, and activity sheets for students in grades K – 8. Download the guides for free at www.chemed.org/ybtc/guides/. CEF’s other programs are the You Be The Chemist Challenge® (a national academic competition for grade 5 – 8 students) and You Be The Chemist® Essential Elements (professional development workshops for educators). In 2014 CEF celebrates its 25th anniversary. Find out more at www.chemed.org.
Related Links
In this Science Friday video, learn how Raul Oaida, at 18 years-old, attached a LEGO shuttle, a video camera, and a GPS tracker to a huge helium balloon and sent them into space:
Want to learn more about spaceflight? Listen to our Q&A with astronauts Don Pettit and Jeffrey Hoffman:
Ask an Astronaut: Don Pettit and Jeff Hoffman on Spaceflight
Education Standards
This lesson applies both Dimension 1: Scientific and Engineering Practices and Dimension 2: Crosscutting Concepts from “A Framework for K–12 Science Education,” established as a guide for the updated National Science Education Standards. In addition, this lesson covers the following Disciplinary Core Ideas from that framework:
PS2.A: Forces and Motion
PS2.C: Stability and Instability in Physical Systems
PS3.C: Relationship Between Energy and Forces
ETS1.A: Definiting and Delimiting an Engineering Problem (see Analysis & Conclusion)
ETS1.B: Developing Possible Solutions (see Analysis & Conclusion)
ETS1.C: Optimizing the Design Solution (see Analysis & Conclusion)
ETS2.A: Interdependence of Science, Engineering, and Technology (see Analysis & Conclusion)
Formulas and Equations
Newton’s laws of motion have played a key role in humans’ understanding of the universe.
- Newton’s First Law of Motion (the Law of Inertia) states: Every object in a state of uniform motion tends to remain in that state of motion unless an external force is applied to it.
- Newton’s Second Law of Motions states: The acceleration (a) of an object as produced by a net force is directly proportional to the magnitude of the net force (F) in the same direction as the net force, and inversely proportional to the mass (m) of the object. This relationship is described by the equation: F = ma.
- Newton’s Third Law of Motion states: For every action, there is an equal and opposite reaction.
- Pressure is the amount of force exerted on an area. This relationship is described by the following equation: p = F/A.
View the original You Be The Chemist® Lesson and Activity Guide – Balloon Rockets and print the activity sheet.
Donate To Science Friday
Make your year-end gift today. Invest in quality science journalism by making a donation to Science Friday.
Meet the Writer
About Chemical Educational Foundation
The Chemical Educational Foundation® is committed to enhancing science education by building greater public understanding and appreciation of chemistry and chemicals through K-8 educational programs and community partnerships. Learn more at www.chemed.org

