Younger Kids Next In Line For COVID-19 Vaccines
12:16 minutes
This story is a part of Science Friday’s coverage on the novel coronavirus, the agent of the disease COVID-19. Listen to experts discuss the spread, outbreak response, and treatment.
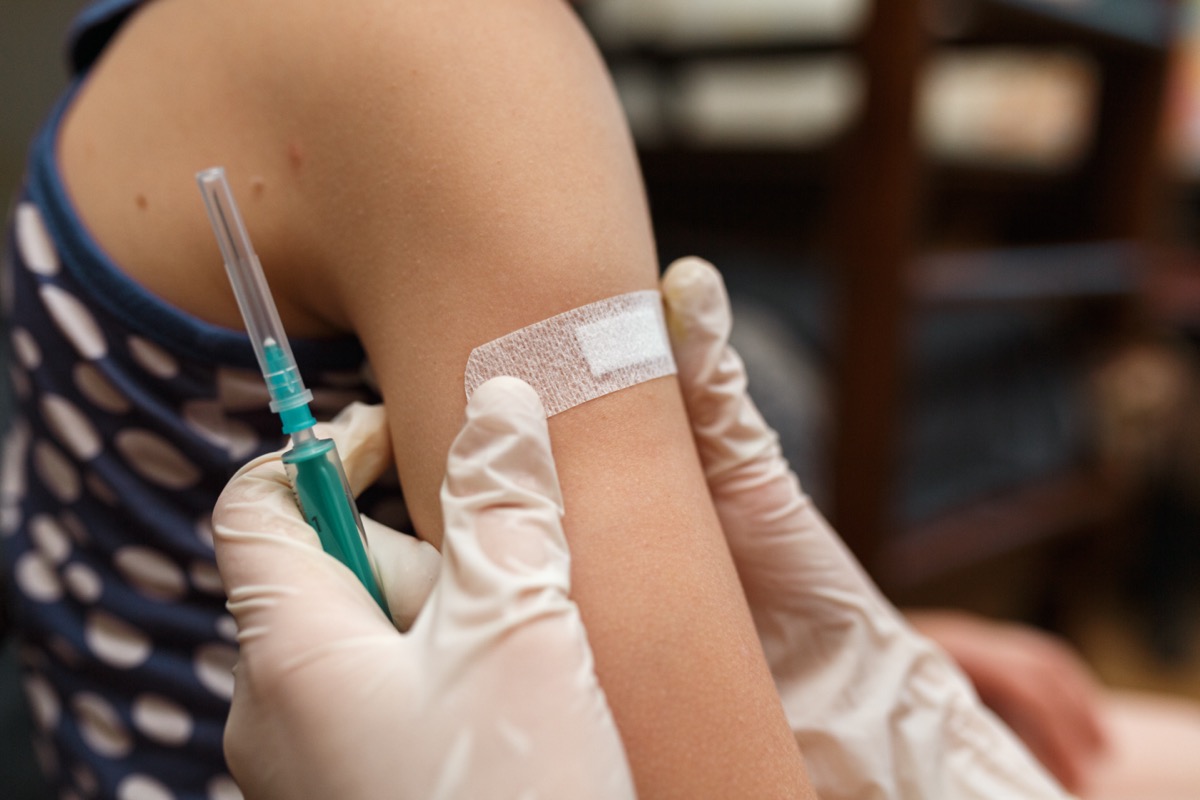
This week, an FDA advisory panel voted unanimously to recommend that the COVID-19 vaccine made by Pfizer vaccine be approved for children as young as 5. If the FDA concurs and the CDC agrees, lower-dose Pfizer vaccinations could soon be available for children ages 5 to 11, via local pediatricians. Just who will be immediately eligible for the doses, and how vaccinating young children might affect school mask policies and other restrictions, remains to be seen.
Umair Irfan, staff writer at Vox, joins Sophie Bushwick to talk about the news and other stories from the week in science, including potential COVID-related criminal charges against Brazil’s Jair Bolsonaro, an experimental bionic vision implant, and the possible discovery of an exoplanet in the galaxy Messier 51.
Invest in quality science journalism by making a donation to Science Friday.
Umair Irfan is a freelance science reporter based in Washington, D.C.
SOPHIE BUSHWICK: This is Science Friday. I’m Sophie Bushwick, technology editor at Scientific American. And this week, I’m sitting in for Ira Flatow. Later in the hour, what biomedical researchers are learning about the medical miracle of maggot therapy. Yes, and ways to listen or watch that you may not expect. Is your potato chip bag giving you away?
But first, progress towards COVID vaccines continues for younger and younger people, with an FDA advisory panel voting unanimously to recommend that the Pfizer vaccine be approved for children as young as five. Vox staff writer Umair Irfan is here with more on that and other stories from this week in science. Welcome back, Umair.
UMAIR IRFAN: Thanks for having me, Sophie.
SOPHIE BUSHWICK: So this week, COVID vaccines for kids got a step closer. Umair, what’s the latest?
UMAIR IRFAN: This advisory committee to the Food and Drug Administration that looks independently at this data basically concluded that the benefits for this vaccine outweigh the risks in ages 5 to 11. This is the Pfizer BioNTech vaccine that already has full FDA approval for adults. And now this moves this to a lower age bracket, which could potentially include upward of 28 million children. And these shots could potentially start rolling out as soon as early next week.
SOPHIE BUSHWICK: Will kids be getting the same dose adults did in the same way that adults did?
UMAIR IRFAN: No, they will not. So one of the critical things they did in this clinical trial is that they used a lower dose. They wanted to measure and ensure that children were still generating the same immune response as adults, but they wanted to use a lower level of the inoculum because they wanted to minimize the amount of side effects that potentially could occur. And a lot of doctors and scientists will tell you, children are not little adults.
And so they wanted to make sure that they started from scratch and made sure they had a dosing that was actually optimal. And so with this one third level dose still administered as two doses spaced three weeks apart, they found that they could generate a similar level of immune response in children, as they did in adults with the full vaccine dose. And they found that the vaccine was 90.7% effective at preventing symptomatic disease in these children.
SOPHIE BUSHWICK: Naturally, some people are going to wonder how much we know about this vaccine in kids. What can you tell us about the risks and the testing that went into it?
UMAIR IRFAN: Well, one thing to consider is that we’ve already distributed this vaccine to hundreds of millions of people in the United States and around the world. And that was a key factor in why these advisors felt very safe in administering this to children. Basically, we know what the potential side effects are. They’re extremely rare. And in the clinical trial, they did not see many of the severe side effects at all in these children. Now this clinical trial that they did for children was much smaller, but again, they felt confident because, again, we have such a huge volume of data to draw on.
Now, the other thing to consider, though, is that children are generally at lower risk for severe disease with COVID-19 compared to adults. And so the cost benefit relationship is a little bit different. That said, we know that children have gotten sick from this disease. Children have died from this disease. And children can transmit this disease.
And so that’s why it’s important to get those children vaccinated to close off that final route of transmission and also why doctors want to make sure that we’re not going to do this through a mass rollout like we did with adults, but rather want children to go to their doctors and clinics and have a physician sort of do the one-on-one talk with them to make sure that they’re perfectly eligible and that this is safe and effective for them in particular.
SOPHIE BUSHWICK: So what happens next? Can we expect vaccine approvals for even younger kids maybe?
UMAIR IRFAN: That is likely the plan. Pfizer says that they are conducting clinical trials in younger groups as well, as young as ages two and also in infants as well. We want to make sure that very young children have weaker immune systems. And so we want to make sure that they can have this level of protection in some of their most vulnerable states and close off the final vulnerable populations and hopefully move us closer towards a more protected society.
SOPHIE BUSHWICK: Moving on to other COVID news, there’s been some potential legal action in Brazil relating to the pandemic. Tell me about that.
UMAIR IRFAN: That’s right. Lawmakers in Brazil, the Brazilian Senate, recommended this week that the president of Brazil, Jair Bolsonaro, be charged with crimes against humanity for his handling of the COVID-19 pandemic. Now those charges will be handed off to Brazil’s prosecutor general, who will have to pursue those charges. It’s not likely that they will. But the fact that the Brazilian Senate actually came to this conclusion is hugely remarkable. They put out this huge thousands of pages report, looking at all the public health strategies that were pursued and weren’t pursued by this government.
For instance, on Bolsonaro’s watch, Brazil suffered one of the worst COVID-19 outbreaks in the world. That resulted in 600,000 Brazilians dying across the country. That’s the second highest death toll in the world, behind the US. Bolsonaro himself got COVID-19. And so there was a whole suite of policies that he did that really exacerbated the situation.
SOPHIE BUSHWICK: So are bad decisions criminal?
UMAIR IRFAN: That’s kind of the thing to consider here. Yeah, we’re in kind of uncharted legal territory. But that’s what the Brazilian Senate is arguing, that even though we have hindsight, they say that at the time, we knew that some of the tactics that the Brazilian government was pursuing were flawed. For instance, Bolsonaro initially ignored the disease and then said that it wasn’t a big deal. He was reluctant to impose restrictions on gathering and implement social distancing measures. Then he promoted hydroxychloroquine, this anti-malaria drug, as a treatment for COVID-19 with very little evidence behind it.
And then he also pursued this herd immunity strategy, where, basically, he thought that letting people deliberately get infected or letting the virus run rampant was a better way to get to widespread protection, rather than actually trying to control the spread. And then a lot of Brazilian senators also said that he botched the vaccine rollout. So a lot of missteps along the way they say stem from a fundamental lack of preparedness and failing to take this pandemic seriously.
SOPHIE BUSHWICK: Is this something he could actually be tried and punished for? Or do you think this is more of a censure in words only?
UMAIR IRFAN: At this point, it does seem like it’s more of a political maneuver. Some members of the Brazilian Senate said they want to submit these charges to the International Criminal Court. And like I said, it’s up to Brazil’s prosecutor general to file charges. And it’s unlikely at this point because they may have some political relationship between them. And so, it’s not likely that he’s going to go to jail.
But again, this investigation this public report that documented, basically, very thoroughly, all the mistakes the Brazilian government made is also sort of a lesson for the next pandemic. This is an interesting public health document as well as a political one that sort of illustrates all the mistakes that were made. We spent a lot of time highlighting how some countries did a good job, but it’s also worth highlighting some of the big mistakes that are out there and what we can do to prevent them the next time another disease rolls out.
SOPHIE BUSHWICK: In better news this week, you have a story about helpful bacteria.
UMAIR IRFAN: Yeah, I was reading a piece in New Scientist by Michael Le Page that looked at this really interesting microbiology experiment. And this was this team that, in a preprint paper, showed that they were able to create artificial symbiotic bacteria. These are bacteria that not just infect cells, but actually live in them harmoniously. And in this case, they can actually produce beneficial proteins.
SOPHIE BUSHWICK: Where did they come from?
UMAIR IRFAN: So this is a bacterium called Bacillus subtilis, which is a common bacteria found in gut. And in this case, they were using them to infect immune cells from mice. And what the scientists found is that the proteins that these bacteria emitted could be used to modify some functions of the cell. And they say that theoretically, you could eventually use these kinds of bacteria to improve healing. You could also improve tissue regeneration, eventually use them to even fight cancer. And the idea is eventually, you can use these modified engineered bacteria as symbiotic helpers for human cells and use them to help treat diseases and other kinds of problems.
SOPHIE BUSHWICK: It seems like if you could get them to release one kind of protein, you might be able to do other sorts as well.
UMAIR IRFAN: That’s exactly right. And we have an example in our own cells already. We have these organelles called mitochondria. And they produce energy for the cell. And some scientists theorize that these organelles actually originated as symbiotic bacteria because these organelles reproduce sort of independently of the cell itself. And so that’s one kind of relationship that’s already occurred in nature.
And they think that if we can cultivate this again, we can do other useful things. Not just for human cells, but also for things like plants. They are saying that you can incorporate nitrogen fixing bacteria directly into plant roots, which can allow them to essentially self-fertilize and help improve agricultural yield, so that’s another potential application.
SOPHIE BUSHWICK: You also have a story this week about bionic glasses. Who are these for?
UMAIR IRFAN: Well, this was a specific case of a 58-year-old biology teacher who lost her sight 16 years ago. In a piece in The Scientist by Lisa Winter, she talked about how this teacher worked with a team at the University of Utah with an implant that went directly into her visual cortex and then connected to a set of glasses. And the research team found that over the course of a few months, they were able to coach this teacher to actually be able to discern letters and shapes.
SOPHIE BUSHWICK: So there’s the glasses part outside, but there’s also a brain implant involved with this. So this isn’t something we’re going to see for casual use, correct?
UMAIR IRFAN: Yeah, this is an experiment, to be clear. And it was temporary. And so the researchers did remove the implant after six months. But it shows that this is a potentially one way to help restore sight. This is somebody who was blind and was able to later see. And now the research team is conducting a clinical trial of this device in four other patients. So potentially, this could be something that is used in some instances to help people regain a sense that they have lost.
SOPHIE BUSHWICK: And lastly, you have a story about a distant planet, but one that’s very, very distant.
UMAIR IRFAN: Yeah, that’s right. A NASA team reported this week that they found the first planet or hints of the first planet for the first time outside of our Milky Way galaxy. This is a Whirlpool Galaxy that’s 28 million light years away from Earth called Messier 51. And they found this using this X-ray observatory. This planet, it’s about as big as Saturn. But it’s about double the distance of Saturn from its star.
SOPHIE BUSHWICK: How do you even start to see something that’s that far away?
UMAIR IRFAN: That’s a really good question. With conventional measures with the way scientists detect planets in our own galaxy, what they do is they look at the light coming off of a star. And they watch for dips in the brightness as planets transit between the star and the observer. And by measuring how much that brightness drops off, they can calculate the size of the planet and how far away it is from the star.
The problem is with visual light, it degrades over very, very long distances. And so you need to look at something that’s more powerful. And X-rays provide that. X-rays can travel over much longer distances. They’re much more energetic. And so by detecting X-rays, they’re able to see these potential planets from much further away. And I keep saying potential because the scientists say they want to verify this by waiting for the planet to transit across the star again. The problem is the next time that’ll happen may not be for another 70 years.
SOPHIE BUSHWICK: So that would be a really long-term experiment.
UMAIR IRFAN: Yeah, we need some very patient scientists or a next generation to sort of pick this up to make sure that they’re on the right track.
SOPHIE BUSHWICK: And how confident are that they’ve got something here? You said they wanted to confirm it, but is that more for peace of mind, or is it something that we should still be unsure about until they’ve got better evidence?
UMAIR IRFAN: They want to make sure that they’re on the right track. They’re using a technique that piggybacks off of existing techniques. But this is the first time it’s been tested. So they want to try this out, looking at other very, very distant stars as well to see if they also have planets. And yeah, it is going to take some time to make sure that this is still a viable technique. But scientists do think that they’re on the right track.
SOPHIE BUSHWICK: And that’s all the time we have. Umair Irfan, staff writer for Vox. Thanks for joining us, Umair.
UMAIR IRFAN: Thanks for having me, Sophie.
Copyright © 2021 Science Friday Initiative. All rights reserved. Science Friday transcripts are produced on a tight deadline by 3Play Media. Fidelity to the original aired/published audio or video file might vary, and text might be updated or amended in the future. For the authoritative record of Science Friday’s programming, please visit the original aired/published recording. For terms of use and more information, visit our policies pages at http://www.sciencefriday.com/about/policies/
As Science Friday’s director and senior producer, Charles Bergquist channels the chaos of a live production studio into something sounding like a radio program. Favorite topics include planetary sciences, chemistry, materials, and shiny things with blinking lights.
Sophie Bushwick is a freelance science journalist and editor based in New York.