Enzymes Are Taking On Our Plastic Problem
9:25 minutes
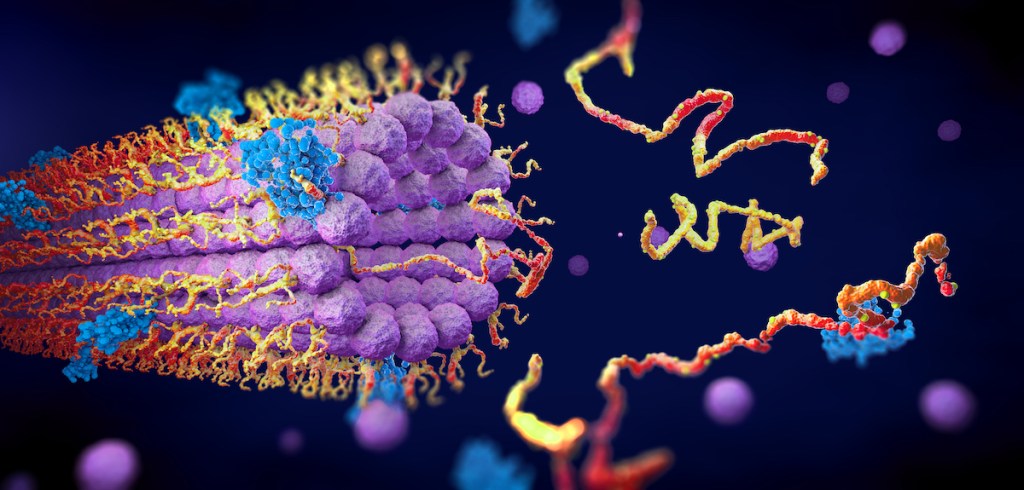
Flip over a plastic water bottle, or a takeout container, and it’s very likely you’ll find the number “1” stamped on the bottom. This is the sign of the problematic plastic PET, which is a large source for plastic pollution. It’s estimated that only a third or less of this type of plastic is recycled into something new.
Scientists are getting creative in trying to outsmart plastics that don’t want to be recycled. Some are looking into enzymes that can break down plastic into its more basic molecular building blocks. The idea is that these smaller molecules are easier to turn into new things, making upcycling an easier task.
Joining Ira to talk about the frontier of enzymes as recycling powerhouses is Jennifer DuBois, professor of chemistry at Montana State University in Bozeman, Montana.
Invest in quality science journalism by making a donation to Science Friday.
Jennifer DuBois is a professor of Chemistry at Montana State University in Bozeman, Montana.
IRA FLATOW: Plastic pollution is everywhere, and it’s a big issue. One problem plastic that’s increasingly common is PET. It’s the very common plastic you find on jars and bottles inscribed with the little number 1 on the bottom. It’s estimated that only a 1/3 or less of this type of plastic is recycled into something new. But scientists are getting very creative in trying to outsmart plastics that refuse to recycle. They’re looking into enzymes that can break down plastic into its most basic molecular building blocks that can be given a second life. And they’re engineering microbes that can make these enzymes.
Joining me today to talk about this new frontier of enzymes as recycling powerhouses is my guest, Jennifer DuBois, professor of chemistry at Montana State University in beautiful Bozeman, Montana. Welcome to Science Friday.
JENNIFER DUBOIS: Hi, there, Ira. Great to be here.
IRA FLATOW: Nice to have you. OK, let’s start with what PET plastic is. Tell us a little bit about it.
JENNIFER DUBOIS: Well, PET is a polymer that consists of alternating subunits. One is ethylene glycol. That’s the diol that’s found in your antifreeze, and the other is terephthalic acid, which we like to call TPA. And TPA is a sort of a flattish, aromatic molecule, if you remember from your organic chemistry days.
IRA FLATOW: My worst subject, my worst. So I’m going to ask you, what makes PET so hard to recycle all these bottles and clothing and carpets that have them?
JENNIFER DUBOIS: Well, the way we recycle it now is really just to chop it up, melt it, and form it into new things. And decoupling those individual monomers from each other is not so hard. It’s just we have to make it really, really cheap. And then we have to find something to do with the subunits once it’s taken apart into the two individual pieces. And right now, the best thing to do with the TPA part is just make more PET.
IRA FLATOW: Hmm.
JENNIFER DUBOIS: And really, what we want to do is get away from materials like PET as much as possible in the future.
IRA FLATOW: So let’s talk about the enzyme that you helped engineer. Tell us a little about what it is and what it does.
JENNIFER DUBOIS: So we’ve been working with actually three enzymes and the first one is kind of the first one out of the gate. And it’s called terephthalic acid dioxygenase. And it takes the terephthalic acid, which, again, is 50% of PET. And inside the cell, it converts the terephthalic acid to an oxygenated product, and with the help of two other enzymes, ultimately makes it into another product called PCA. Sorry for the alphabet soup.
IRA FLATOW: Yeah, and so what happens with the products that are done, that you’re left with?
JENNIFER DUBOIS: So one reason why we love PCA as a product, instead of TPA, is that we want to be able to convert lots and lots of materials, be they natural or non-natural, into central compounds that we can use for something else. So if we take something complicated like a tree or something chemically more simple like PET and we grind it all up, ideally, we want to have just one or two things come out the funnel at the bottom. Those are things that we can collect and give to chemists to find ways to use. And PCA is something that is a pretty great funneling compound.
IRA FLATOW: And so what do you get when that happens?
JENNIFER DUBOIS: Well, PCA itself, right now, a lot of scientists are working on finding, basically, stuff to do with it. And inside of a microbe, the microbe can be sort of re-engineered to take that PCA and convert it back into a form of biological nylon. We could just feed a bunch of bacteria, our polyethylene terephthalate, which we ultimately got from petroleum, and then they would convert it to carbon dioxide and water.
And at least, we would have gotten rid of some garbage, but we would have put it right up into the atmosphere. But with upcycling, what we really want to do is collect the carbon from that polyethylene terephthalate and change it into something valuable. And so now we have at least one destination for PCA that could be carried out fully within a microbe. And that is to re-convert it into a form of nylon. But the sky is sort of the limit with what you can do with a compound if you have a creative chemist stare at it for a while.
IRA FLATOW: If there are so many tons and tons of the PET plastic around, and there are microbes that would love to eat it and can be engineered to do that and create products you want, why aren’t we seeing more of these plastics being eaten by these microbes?
JENNIFER DUBOIS: That’s a great question. And I think what we’re seeing is something like evolution occurring in real-time. Terephthalic acid is not really an abundant natural product. And it’s something that these bacteria have been exposed to quite a bit, especially bacteria and wastewater treatment and other contaminated sites. And so they take an existing set of their own enzymes, and we call this evolution by duplication. They duplicate them. And then the duplicate evolves to handle a brand new compound that these organisms are now being subjected to having to deal with.
And so what we think is that some of the enzymes and metabolic pathways that we’re discovering are really at the threshold of evolution, and they’re not great. And that is probably why we don’t see them, really, all over the place.
IRA FLATOW: This is Science Friday from WNYC Studios. In case you’re just joining us, we’re talking to Dr. Jennifer DuBois, professor of chemistry at Montana State University, about using bacteria that make enzymes to recycle plastics. So is it possible that a recycling plant for plastics may actually turn into a place where you feed these plastics to the bacteria who chomp on them and make them disappear?
JENNIFER DUBOIS: That’s just a beautiful vision of the future, isn’t it?
IRA FLATOW: Yeah. You’re telling me it couldn’t happen?
JENNIFER DUBOIS: I think that we sort of feel like if it’s going to happen, PET, or PET, is the material that it’s going to happen for first. PET looks a lot like a natural product in many ways. And so, in some ways, it’s not surprising that bacteria figured out how to eat it. So we think of all the plastics, and like you mentioned, this is plastic number one. PET is really the front runner for biological upcycling. But we still have five other plastics. And what we imagine is a future where those other plastics may be converted by biological or other means. So this is definitely a multifaceted problem.
IRA FLATOW: Yeah. It’s Earth Day. And in some ways, it seems appropriate and a little ironic that some of the smallest things on Earth are helping to solve one of the biggest problems we humans make.
JENNIFER DUBOIS: I think it’s beautiful.
IRA FLATOW: Yeah, I hope it comes to fruition.
JENNIFER DUBOIS: Well, there’s definitely a lot of us working really hard to make that vision a reality. And these are projects that just light the fire underneath the feet of young people. I have two outstanding grad students, Jessica and Rita, who are on fire to do this work. And I think young people are just so engaged with it.
IRA FLATOW: Well, we’ll leave it there and hope that the young people are the key to our future, which I’m counting on in a lot of different ways when it comes to the Earth.
JENNIFER DUBOIS: It was my pleasure. Thank you.
IRA FLATOW: Dr. Jennifer DuBois, professor of chemistry at Montana State University in Bozeman, Montana.
DIANA MONTANO: I’m Diana Montano with another meditation for your Earth Day. And this time, I’ve got a true, false question for you about one of our favorite subjects– cephalopods, like octopuses and squids. I’ll read the question and give you a few moments before I tell you the answer. Ready? Here we go. True or false? Although they are masters of camouflage, researchers believe cephalopods are colorblind. Their eyes can only see in black and white.
Well, it’s true. Cephalopods can’t see color, at least not with their eyes and not the same way that we see color. We still need more research to understand exactly how they mimic their environments so closely. Until then, keep a keen eye out for an octopus during your next snorkeling trip. If you love trivia questions about the Earth and want to join me for our weekly trivia nights, go to sciencefriday.com/trivia.
IRA FLATOW: Thank you, Diana. Diana Montano, SciFri’s trivia night host.
Copyright © 2022 Science Friday Initiative. All rights reserved. Science Friday transcripts are produced on a tight deadline by 3Play Media. Fidelity to the original aired/published audio or video file might vary, and text might be updated or amended in the future. For the authoritative record of Science Friday’s programming, please visit the original aired/published recording. For terms of use and more information, visit our policies pages at http://www.sciencefriday.com/about/policies/
As Science Friday’s director and senior producer, Charles Bergquist channels the chaos of a live production studio into something sounding like a radio program. Favorite topics include planetary sciences, chemistry, materials, and shiny things with blinking lights.
Kathleen Davis is a producer and fill-in host at Science Friday, which means she spends her weeks researching, writing, editing, and sometimes talking into a microphone. She’s always eager to talk about freshwater lakes and Coney Island diners.
Ira Flatow is the founder and host of Science Friday. His green thumb has revived many an office plant at death’s door.