Subscribe to Science Friday
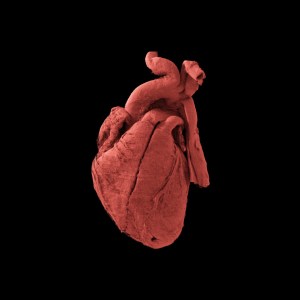
A new imaging technique using a particle accelerator is giving researchers an unprecedented level of detail of our organs, producing scans 100 billion times brighter than a CT scanner. Those 3D models are now part of a public database called the Human Organ Atlas, available to researchers and the medically curious.
Joining Host Ira Flatow to explain why they needed so much power and what kind of research advances will follow is imaging scientist Claire Walsh, director of the Human Organ Atlas hub.
Images from the Human Organ Atlas
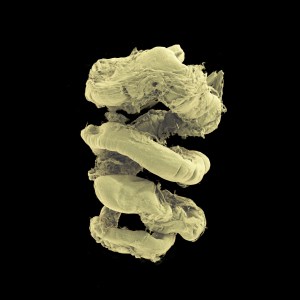
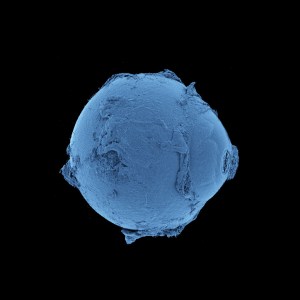
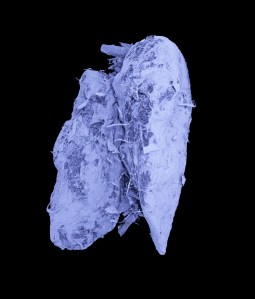
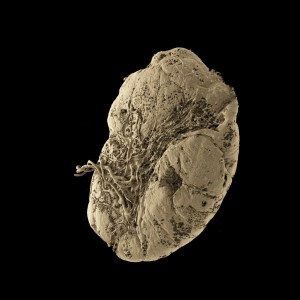
Donate To Science Friday
Invest in quality science journalism by making a donation to Science Friday.
Segment Guests
Dr. Claire Walsh is an associate professor at the UCL department of mechanical engineering and director of the Human Organ Atlas Hub.
Segment Transcript
[MUSIC PLAYING] IRA FLATOW: Hey, it’s Ira. And you’re listening to Science Friday. When I was a kid, my dad gave me a copy of Gray’s Anatomy, that 5-inch-thick classic anatomy textbook, and I became fascinated with the human body. I loved going over the hyperdetailed illustrations of our muscles and our organs. And ever since then, I’ve been a fan of unique anatomy books.
Well, my next guest is leading a project that brings Gray’s Anatomy into the digital age. It’s called the Human Organ Atlas. It’s a new public database of organs that are scanned with the help of an actual particle accelerator, yeah, giving visitors an up-close and personal look at 56 human organs. Here to explain why they needed so much power and what kind of research advances will follow is Dr. Claire Walsh, associate professor at University College London and Director of the Human Organ Atlas Hub. She researches 3D imaging techniques for human organs. Welcome to Science Friday.
CLAIRE WALSH: Hi, Ira. Thank you very much for having me here. It’s a pleasure.
IRA FLATOW: You’re welcome. I’ve got to say, these organ images, they’re just stunning. You can see tiny, individual ridges on the eyeball, all the tiny, little pockmarks in the lungs. What was it like for you seeing these for the first time?
CLAIRE WALSH: Yeah, I think, I mean, as an imaging scientist, images is what I love to work with and to deal with. And I think, going all the way back to 2021, when we first started to get these images through, it was really a wow moment, the first time we saw the images of the lung, which was what we started with, coming off the particle accelerator that we use, because this was all during the COVID pandemic, opening them up, doing screenshares with all the collaborators on the projects there, and everyone just sort of taking a minute, taking it in, and being like, wow, what are we even looking at? This is amazing.
Anyone can explore it. You’ve got explore and search functions. And you can click through the different organs. You can look at the pictures. And most importantly, you can explore the actual real datasets yourself online there. We use a viewing portal on the website, very similar to what powers Google Satellite or Google Earth. So you can basically open the datasets up in the little window there, and you can look through them. And you can zoom in to different places, and it pulls into higher resolution. And you can explore the 3D space of an organ, which is very cool.
IRA FLATOW: So Google Earth for your organ.
CLAIRE WALSH: Exactly, yeah.
IRA FLATOW: So this started during COVID. This wasn’t a project, say, hey, we’re all sitting around, let’s do something different?
CLAIRE WALSH: I mean, so the very back inception of this project was during the COVID pandemic in 2021, and it was actually started because some medical collaborators of ours from the Hanover Medical Center were looking for ways to better understand the lungs of the COVID-19 patients that they were seeing, and it was in the thick of the pandemic.
And they got in contact with Professor Peter Lee, who’s here at UCL. And he was like, I know person we need to call. He called Paul Tafforeau, who is at the Particle Accelerator in Europe. And together, we kind of all pushed the technology together to answer these questions of what’s COVID-19 doing to the lungs of these people. But very soon after that, we realized, hey, this technique will be amazing, not just for lungs, but for all the other organs of the human body as well.
IRA FLATOW: Now, you’re borrowing a big, basically atom smasher that physicists use to look for subatomic particles. I mean, how do they feel about you taking over their machine?
CLAIRE WALSH: [LAUGHS] Well, so we’re similar kind of ideas. So some of the really big particle accelerators, like the CERN, the Large Hadron Collider in CERN, which a lot of people have heard about– it’s actually kind of just up the road from the particle accelerator we use, Geneva and Grenoble.
And the synchrotrons, they’re slightly different to something like the Large Hadron Collider. The Large Hadron Collider is accelerating these particles and smashing them together. With the synchrotron, you’re accelerating these electrons around, and you’re not smashing any of them together. You’re just bending them, and you’re using the radiation that comes off.
IRA FLATOW: Aha.
CLAIRE WALSH: So the synchrotron, they do a lot of different things, but imaging is a big part of what many of these synchrotrons do. They’re not just imaging human biomedical features, it should be said. They have a huge range of things that they do. Crystallography is a big part of what some synchrotrons do. They’re also using the similar technique, but they’re imaging things like car parts and aeroplane parts and batteries and lots of incredibly exciting research.
IRA FLATOW: And now hearts and kidneys and things like that.
CLAIRE WALSH: Exactly, yes.
IRA FLATOW: Oh, you raised so many questions I need to ask. First of all, whose organs are these?
CLAIRE WALSH: Yeah, so I think it’s really important for everyone to understand and to see these are all organs that we’re imaging of people who have died and donated their bodies to science. None of this imaging is done in live human beings. The X-ray dose would be far too great.
IRA FLATOW: Why can’t you just do, let’s say, a CT scan? This sounds like hitting it with a hammer. Tell me about that.
CLAIRE WALSH: Yeah, so the imaging technique, it’s called HiP-CT, Hierarchical Phase Contrast Tomography. It doesn’t have anything to do with hip joints. That’s just the acronym.
IRA FLATOW: [CHUCKLES]
CLAIRE WALSH: But essentially, you can kind of think of it like a supercharged CT, in a way. The kind of underlying principles are the same. You take an organ and you take an X-ray beam, and you shine that X-ray beam through the organs and you rotate them around slowly. And you’re taking images at all these different rotation angles, which is the same way that a hospital’s CT scanner works. But there’s a few really critical differences.
One is the types of X-rays that we’re using. So we go all the way to a particle accelerator in Grenoble, France, called the European Synchrotron Radiation Facility. And what this particle accelerator does is accelerates electrons around a ring that’s about 800 meters in circumference, and it accelerates them up to nearly the speed of light.
Now, when you accelerate electrons that fast, and you bend them around like a ring around a circle, they emit radiation as they bend. It’s called synchrotron radiation. It’s wavelengths of X-rays. And it’s that that we’re using to image the samples. And this X-ray is different to the X-ray that you get in hospitals because it’s much, much more– it’s called brilliant. It’s a more brilliant source, which basically means it’s much more intensely focused and much brighter than the X-rays that you would get in a hospital CT scanner.
So when you have these way, way more brilliant and higher energy and higher power kind of X-rays, you can do some interesting things that you couldn’t do with a hospital. The first thing is that if you go into a normal hospital CT scan or an X-ray scan, you’re shining X-rays through the tissue. And the image that they get on the other side is because some of the tissues in your body absorb X-rays more than others.
So like, bone absorbs a lot of X-rays. So if you’re in an X-ray and there’s bone in there, the X-rays will be absorbed by the bone, but they’ll pass through the softer tissue more. And so that’s why the bones appear kind of bright, and the softer tissue appears. And it’s the same thing with a normal CT. The difference that we have here is that as the X-rays pass through the tissue, we’re not as interested in how much the signal is absorbed. That is a factor. But we’re actually interested in something else that happens to X-rays.
So as X-rays pass through tissue, X-rays are waves, if you’ll imagine. And these waves are shifted. As they pass through tissues with different densities, the waves are shifted relative to one another. And when these waves get shifted relative to one another, you get what’s called an interference pattern. And it’s this interference pattern that we can measure, and it basically gives us the ability to look at really small differences in density between the tissue. And so those are the primary differences that you get. You couldn’t do this in a hospital CT scanner.
IRA FLATOW: Yeah, you’re getting a lot more detail, right?
CLAIRE WALSH: Exactly. And we’re at a much, much higher resolution. So a normal hospital CT scanner, you’re kind of 1 millimeter size for each of the little cubes that make up the image. So the resolution, you can say, is like 1 millimeter. With the HiP-CT technique, we go down to 1 micrometer.
IRA FLATOW: Wow.
CLAIRE WALSH: So you’re like 100th the diameter of a human hair.
IRA FLATOW: So that’s got to be a big file size for that data.
CLAIRE WALSH: Really, really big data set. So our data sets are kind of terabytes in size for a lot of the data. And that’s a big part of the Human Organ Atlas that we’ve created, is how do you make data that size available to anybody around the world, and not just available, but how can you allow them to interact with it in a way that’s interesting and meaningful, like scientifically and educationally?
IRA FLATOW: And tell me why this is so useful for researchers to have super detailed scans of organs?
CLAIRE WALSH: Yeah, so there’s a whole bunch of ways that these scans are really useful for researchers. And some of it is in terms of just medical education and understanding anatomy. So that’s one idea, anatomy researchers who are trying to understand, OK, where does this anatomical detail fit? We’re seeing for the first time here, in undistorted three dimensions, what anatomical features look like.
If you were to do physical anatomy dissection, some structures you won’t be able to see because they’re too small. And also, you have to cut an organ up to get to the internal structure. And sometimes if you’ve made a cut in one direction, you can’t see a structure. Maybe you’ve cut through it, or you’ve cut at an angle that doesn’t allow you to see that structure. With this kind of virtual, digital version of the organ, you can cut in any direction you want, and you can see very small structures. So that’s one thing.
Another big area that we look at and we work at is we do AI-assisted segmentation of structures. So one that we’ve worked on a lot is the blood vessels. So looking at the blood vessel network in the human kidney, for example. And with these data sets, we have a load of human kidneys within the Human Organ Atlas, and you can extract the blood vessel network across all these kidneys.
And you can say, hey, how does the blood vessel network change between male and female kidneys? Or how does it change in the younger versus the older kidneys? Or how is it changed by donors where they have conditions like diabetes in them? We can look at these kind of differences and understand how does that change its structure.
IRA FLATOW: Hmm. Say more on this, because I want to know perhaps what are some other ways that the AI machine learning could come into this.
CLAIRE WALSH: Yeah, so I think there’s tons of areas that are really interesting that we’re looking at with the AI and the machine learning. A lot of it is around looking at different structures of interest. So the blood vessels is one that I’ve mentioned, but we also have projects in the human brain. And we’re looking at the connectivity of the white matter within the human brain. So the white matter in the human brain is kind of the wiring portions of the brain that you can imagine.
And with HiP-CT, we can look– those wires are actually individual axons, cells with very, very long tails that join parts of the brain to other parts of the brain. With the HiP-CT, you can look at the orientations and the directions that these connections are going. And you can start to unpick, OK, what’s the wiring diagram of the human brain? So that’s another project that we work on with this data.
We also have really exciting work going on in the heart, looking at when you have congenital heart disease, how the structures within the heart change when you have these kind of congenital diseases. Structures that we expect to be in one location are actually maybe in a different location. And if you’re a surgeon performing– if you’re a surgeon doing a surgery on a condition to repair congenital heart disease, it’s really useful to know, OK, where might these structures actually be in this kind of different heart?
IRA FLATOW: We have to take a quick break, but don’t go away. More on this when we come back.
[MUSIC PLAYING]
[MUSIC PLAYING]
IRA FLATOW: Oh, so that’s how this can filter down to actual patients.
CLAIRE WALSH: Yeah, that’s one of the ways in which it can filter down to actual patients and training, surgical training for surgeons to understand better the anatomy and then to improve their surgical skills. But there’s also some other really interesting ways that are a bit more long-term that we’re looking at where we look at these very high resolution HiP-CT scans, and we compare them to clinical scans, so like a clinical MRI or a clinical CT scan, that this donor may have had when they were still alive.
And by comparing those scans to each other, it’s another area that AI can come in, where you say, OK, can we learn anything from this very, very high-resolution image that we have from HiP-CT? Can we compare it to the scans the donor had when they were in life? And are there any things that we can see in that clinical scan that we didn’t really know were there? But actually, once you have the HiP-CT data, and you align those two, can you suddenly see other features that you maybe weren’t able to extract from the original clinical data?
IRA FLATOW: Now, I know that since you can only scan cadavers, I’m sure there are certain medical conditions you couldn’t use this approach with. I mean, tell us about maybe how you might get around that or how you could use that.
CLAIRE WALSH: Yeah, so I mean, the other thing about HiP-CT is that it’s a static technique. The organs are fixed in place. And at the moment, we can only scan them like that still. So there’s a lot of conditions. Human bodies are not static statues. They’re dynamic systems. And a lot of times, some of the conditions of interest and the problems that we have are to do with that more dynamic part.
So any conditions where something that’s kind of– something that’s dynamic or is changing that doesn’t result in a structural change to the organ is going to be kind of invisible to HiP-CT. You can imagine, for example, very simply, kind of like a leukemia, like a blood cancer. Like, HiP-CT, we’re not imaging the flow of the blood and things. And where that’s the primary issue, you don’t really have access to that with the HiP-CT as much.
IRA FLATOW: Do you think you could ever eventually scan a whole body?
CLAIRE WALSH: Yeah, I think, that’s definitely a goal more long-term in the next five years, is to be able to scan an intact, whole human cadaver. And I think that would just be a really scientifically interesting thing to do because at the moment, we’re scanning individual organs, but again, human body’s organs don’t work individually on their own in a body. They work as part of a system. Like, the heart and lung, they function together as a system.
And so when you image one separate from the other, you lose a load of that connectivity between the two. That’s incredibly important in terms of how they actually function. So by scanning a whole, intact human body, you preserve those connections between the different organs, and you get a much better holistic understanding of how does the body function and how does that go wrong in disease.
IRA FLATOW: I know you said you were really surprised when you first saw the– what was it– the lungs, right? Do you have a favorite organ? Or is that your favorite?
CLAIRE WALSH: So I think my– personally, I work a lot with the kidneys and the brain. And one of my favorites is actually the kidney. I think it’s a bit of an unsung hero. It’s a beautiful organ to look at. It’s kind of under-recognized, I think, maybe. But it’s really interesting, fascinating, how these different compartments within the kidney that serve these very different roles.
And I think, also, in terms of understanding human disease, kidney disease is a huge burden on health care systems worldwide. And I think this technology offers such a powerful tool for understanding that and maybe being able to mitigate some of the problems people have with transplant rejection, chronic kidney disease, and a whole range of other things.
IRA FLATOW: Well, this has certainly been fascinating stuff. I want to thank you for taking time to be with us today. And good luck to you in the future.
CLAIRE WALSH: Thank you very much, Ira.
IRA FLATOW: Dr. Claire Walsh, director of the Human Organ Atlas Hub. And you can check out more of these stunning images. They’re on our website at sciencefriday.com/organs. Speaking of body parts, we got this call from a listener I think you’ll enjoy. We did a segment about the uses of spider silk a few weeks back, including some of its healing properties, and got this call.
LINDA: Hi, it’s Linda, calling from New Jersey. I was listening to the program about spiders and wanted to let you know that my grandmother, who was born in 1889, Kamionka Strumiłowa, Poland, told me that when she was a girl and she cut her hand very badly, she put it in a spider web, and it stopped the bleeding. And it helped the cut to close up. And she always believed in using spider webs to heal things. So just wanted to let you know that. Thanks for all the information. Bye.
IRA FLATOW: Thanks for calling, Linda. This episode was produced by Dee Peterschmidt. I’m Ira Flatow. Thanks for listening.
[MUSIC PLAYING]
Copyright © 2026 Science Friday Initiative. All rights reserved. Science Friday transcripts are produced on a tight deadline by 3Play Media. Fidelity to the original aired/published audio or video file might vary, and text might be updated or amended in the future. For the authoritative record of Science Friday’s programming, please visit the original aired/published recording. For terms of use and more information, visit our policies pages at http://www.sciencefriday.com/about/policies/
Meet the Producers and Host
About Ira Flatow
Ira Flatow is the founder and host of Science Friday. His green thumb has revived many an office plant at death’s door.
About Dee Peterschmidt
Dee Peterschmidt is Science Friday’s audio production manager, hosted the podcast Universe of Art, and composes music for Science Friday’s podcasts. Their D&D character is a clumsy bard named Chip Chap Chopman.