The Not So Inactive Ingredients In Our Most Common Drugs
7:39 minutes
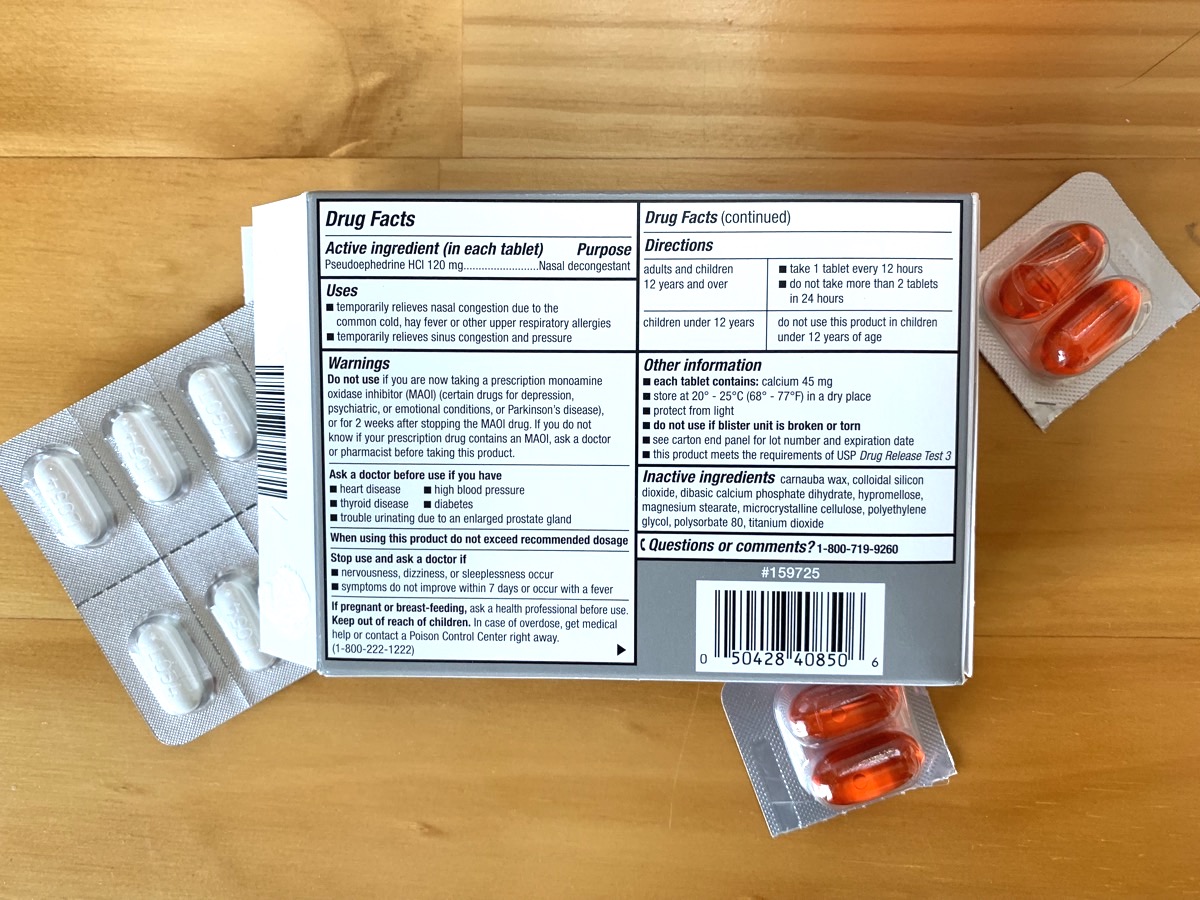
Anyone who has glanced at the back of a bottle of aspirin or a box of allergy tablets has seen it: the “Inactive Ingredients” list. All medications include compounds that help stabilize the drug or aid in its absorption. They aren’t given a second thought because they’re “inactive,” which suggests that these ingredients don’t do any harm.
But in fact, according to a new study out this week, over 90 percent of medications have inactive ingredients that can cause allergic reactions in certain patients, including peanut oil, lactose, and gluten.
Dr. Giovanni Traverso, an gastroenterologist at Brigham Women’s Hospital, and an assistant professor at Harvard Medical School and MIT’s Department of Mechanical Engineering, joins Ira to share the not so inactive ingredients in some of our most common drugs.
Credit: Diana Saville
Giovanni Traverso is a gastroenterologist and biomedical engineer at Brigham and Women’s Hospital and Harvard Medical School, and a research affiliate at MIT in Cambridge, Massachusetts.
IRA FLATOW: This is Science Friday. I’m Ira Flatow. Inactive ingredients. You’ve seen them on the back of that bottle of aspirin you have at home or a box of allergy medication you’re purchasing. Your eyes zoom right by them as you’re looking for the dosage or list of side effects.
You don’t give them a second thought because of what they’re called, inactive ingredients. That means they don’t do anything, right? Well, look a little bit closely at those inactive ingredients, and you’ll find they include things like peanut oil, lactose, and gluten.
And in fact, there’s a new study out this week shows over 90% of medications have inactive ingredients that are really active, that can cause allergic reactions in certain patients.
Here to tell us what to make of all of this is one of the authors of that study, Dr. Giovanni Traverso, assistant professor at Brigham Women’s Hospital, Harvard Med School, and the Department of Mechanical Engineering at MIT. Dr. Traverso, welcome back to Science Friday
GIOVANNI TRAVERSO: Oh, thanks so much, Ira, for having me.
IRA FLATOW: So what else does my blood pressure medication– not that I take it–
[LAUGHS]
–Doing for me besides the medicine itself? What kind of compounds are you finding in these inactive ingredients?
GIOVANNI TRAVERSO: I mean, I think it’s really fascinating what we find, and this was sort of something that I came across a few years ago. And you know, one of the things that we set out to do in the study is really analyze exactly what else is in there.
And you know, on average we find that about 75% of tablets and capsules are actually occupied by these inactive ingredients. And typically, on average, there are about eight other ingredients in that capsule and sometimes up to 35.
And as you mentioned earlier, they’re ingredients like lactose, sometimes starch, which can be wheat derived. And so hence, the potential for gluten. In some instances, also even peanut oil. But many other– there are over 1,000 chemicals that one can choose from to really help make that capsule.
IRA FLATOW: So why did the pharmaceutical companies, if they know this kind of stuff– I mean, they know that people are allergic to lactose, and wheat, and peanuts. Why do they put them inside the pills?
GIOVANNI TRAVERSO: Yeah. So I think one thing to really emphasize here is that these inactive ingredients are actually really important. So I think by no means that we are suggesting that they be removed. And they play a critical role with respect to the stability of that tablet or capsule, the appearance, potentially modulating taste, or even enhancing absorption, or even preventing tamper proofing.
And you know, likely, the reason that we have some of these ingredients is for historical reasons, where we’ve seen some of these work to actually facilitate those parameters in the pills.
But certainly as awareness increases, it’s something that I think we’re all starting to appreciate, hopefully, more and more. And I think hopefully we’re getting more focus around what should be included, and can we find alternatives in some situations
IRA FLATOW: And you think that there are alternatives that could used?
GIOVANNI TRAVERSO: Absolutely. You know, and if you take drugs, drugs that are now generic– for example, omeprazole, which is used for the treatment of reflux or ulcer treatments. There are many different formulations of omeprazole.
So for example, the physician– like, I will prescribe, let’s say, omeprazole 20 milligrams once a day or twice a day. But at that dose, there can be over 30 or 40 different formulations of omeprazole 20 milligrams.
And by formulation, I mean the composition of the inactive ingredients. So one might find ones that have lower amounts of lactose or no lactose and others that may have lactose. And so, you know, certainly there are alternatives. And for some drugs, there are many alternatives. For others, there are a few.
IRA FLATOW: You know, sometimes when I go to the drugstore and I have a medication that I’m getting. And the druggist says, you know, a different manufacturer’s making that same drug, and we’re using that now. That could have other ingredients in it that I’m not used to from the first pharmaceutical company.
GIOVANNI TRAVERSO: That’s exactly right. And I think, myself included and colleagues, you know, we’ve seen situations where– similar experiences that our patients have had, where, exactly as you mentioned, the manufacturer changes.
IRA FLATOW: Yeah.
GIOVANNI TRAVERSO: And actually, they’re manifesting actually new symptoms, potentially. And I think that’s– hopefully by raising awareness that there are inactive ingredients in these pills and capsules, that they may be the culprit behind some of those symptoms.
IRA FLATOW: Speaking of your colleagues, when you found out that 90% of the medications had these ingredients in them, what was the reactions from your prescribing colleagues to this news?
GIOVANNI TRAVERSO: So I think, though, you know, just to take a step back on the 90% piece, one of the things that we recognize of these inactive ingredients is that they can cause adverse effects. And generally, we group them into two groups. One is allergic reactions, so those sort of frank allergies, as we recognize them today, whether it be in the extreme, like an anaphylactic reaction or a rash.
And then on the other side is sort of intolerances, like GI intolerances. On sort of the allergy side, what we had identified in this study from the literature is that there are 38 ingredients have been associated with allergies.
And if one asks the question, what percentage of capsules contain at least one, the answer is indeed over 90%. But I think one thing to emphasize is that, you know, those events are rare. But nevertheless, they are out there, but they are rare.
So I think people still recognize that these events are rare. But you know, I think just– again, I think it’s all about raising awareness, both at the patient level and at the health care provider level. And I think, you know, it’s changing. I think, how we think about the prescription process and access to the information.
IRA FLATOW: So let’s say I’m a concerned consumer. Is there any place online that I can check out what I should be worried about? Is there a resource?
GIOVANNI TRAVERSO: Absolutely. So I think you mentioned at the beginning, certainly the drug inserts. So you could sift through the text and you will find some of that information. There’s also a database out there called Pillbox that has some of that information. But for both, there are a few steps to go through to at least arrive at that.
So one of the things that we see as potentially helping both patients and health care providers is developing tools to just make it easier to really help identify those ingredients very quickly and even to help quantify the total amount of an ingredient.
So let’s go back to lactose. If someone is taking, let’s say, 10 tablets, to at least quickly figure out how much lactose that person is consuming from their medications alone. And so that’s one of the next things that we’re starting to look at.
IRA FLATOW: You say developing tools. What do you mean? Like an app or something like that?
GIOVANNI TRAVERSO: Exactly. I think it’s apps. It’s also systems to interface with the electronic medical record systems to really start to help quantify the inactive ingredient components.
IRA FLATOW: Interesting. Thank you for taking time to be with us today, Dr. Traverso.
GIOVANNI TRAVERSO: Well, thank you, Ira, for having me on the show.
IRA FLATOW: You’re welcome. Have a good weekend.
GIOVANNI TRAVERSO: Likewise.
IRA FLATOW: Giovanni Traverso is a gastroenterologist at Brigham and Women’s Hospital, assistant professor in the Department of Mechanical Engineering at MIT. So he works both MIT and Harvard at the same time.
Copyright © 2019 Science Friday Initiative. All rights reserved. Science Friday transcripts are produced on a tight deadline by 3Play Media. Fidelity to the original aired/published audio or video file might vary, and text might be updated or amended in the future. For the authoritative record of Science Friday’s programming, please visit the original aired/published recording. For terms of use and more information, visit our policies pages at http://www.sciencefriday.com/about/policies/
Katie Feather is a former SciFri producer and the proud mother of two cats, Charleigh and Sadie.
Ira Flatow is the founder and host of Science Friday. His green thumb has revived many an office plant at death’s door.