Grade Level
9-12
minutes
1- 2 hrs
subject
Life Science
Activity Type:
build a model, protein folding, structure and function, biodiversity and adaptation
Picture the common garden snail, peacefully oozing along a branch to find some leaves to nibble on. But it’s a much different picture with some marine snails, like the cone snail. This seemingly innocuous creature is designed to kill, quickly striking its prey and eating an entire fish in one gulp.

Intrigued? Learn from researchers as they document “killer snails” in action and study the proteins that these predatory snails use to subdue their prey. You’re going to see how cone snails hunt prey in the wild, create your own snail model to visualize the structures they use for predation, and study some of the peptide proteins used by snails to paralyze their prey.
So, put on your wetsuit, mask, and flippers, grab your waterproof camera, and get ready to explore!
What Does It Look Like When Predatory Marine Snails Attack Their Prey?
If you were to dive into the water to observe killer snails, here’s what you might see through your snorkel mask. As you watch, try to observe the sequence of events leading to the capture and consumption of fish prey:
Share Your Observations: Describe three moments of the interaction you observed between the snail and the fish for sharing on social media. Try to describe what happened at the start, middle, and end of the interaction, thinking about how you can accurately represent the predation event using words and illustrations in a way that will earn as many likes and shares as possible. Use this social media worksheet to draft your posts.
Meet A Killer Cone Snail

You’ve just encountered a cone snail, Conus magus, also called the “magical cone.” Though they can go unnoticed as they sit silently beneath the sand, they can pack quite a punch. As you just observed, in less than 30 seconds, the magical cone can harpoon, tether, paralyze, and engulf an unsuspecting meal. Giving new meaning to the expression, “going at a snail’s pace”!
Of the more than 700 different species of cone snails, all of them are venomous predators, and all share a set of essential structures for capturing, poisoning, and consuming their prey. Here’s a closer look at the weaponry cone snails use to capture and kill their prey:
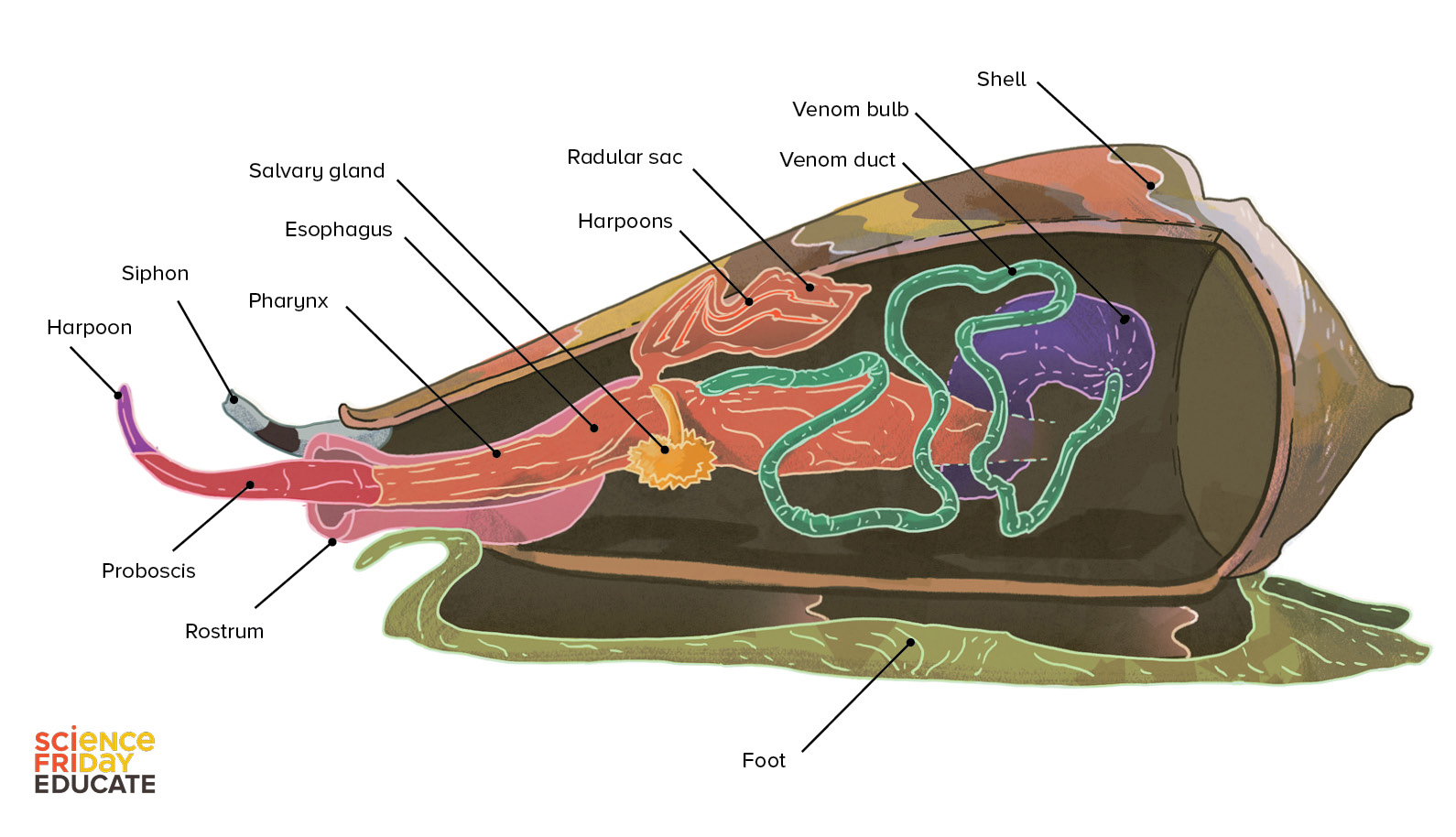
Each structure has a special function that helps a snail hide, pounce, kill, and consume its prey.
Like an undercover assassin, a cone snail uses its muscular foot to burrow, conceal, and anchor itself to a rock as it patiently awaits its victim. Like a nose, the siphon sniffs out prey. Like protective armor, its tough calcium carbonate shell safeguards it from larger, hungry predators.
What’s on the menu? Depending upon the species, everything from fish to marine worms to even other snails. The proboscis stretches out like a tongue, tipped with a venom-coated harpoon to stun, paralyze, and capture its prey. The venom bulb acts like a pump to deliver venom through the venom duct into the harpoon, injecting it into the prey. The rostrum acts like a mouth, engulfing the prey in one big chomp. Some hours later, the snail burps out any indigestible fish parts such as scales and fish bones, along with one used harpoon. Before its next hunt, another harpoon will be retrieved from its radular sac and reloaded into the proboscis ready to stun its next prey.
Make A Cone Snail Model

Use the description of the anatomy and feeding behavior of a cone snail (above) to guide you as you build a working model of a cone snail. In your model, be sure to include all of the cone snail structures needed to capture a fish (in bold). The structures that you model will need to be functional enough to demonstrate how cone snails kill their prey.
The following are suggested materials only–get creative! You can use any materials you’d like to model snail feeding structures and their functions.
Suggested Materials
- Shell: Plastic container, paper cup, or other container
- Siphon: Pipe cleaners, elastic bands, small straws, stir sticks
- Proboscis: large or small bendy straws
- Harpoons: Plastic cocktail sword, straight pin, or round toothpick
- Venom: Colored beads, pom poms, or food sprinkles
- Venom bulb: balloon, paper cup, ping pong ball
- Venom duct: paper straws, plastic tubing
- Prey: Swedish gummy fish or gummy prey
- Muscular foot: paper, spare socks,
- Rostrum: Spare socks, balloons, etc.
- Radular sac: Small paper cup, balloon, baggie
- Your hands!
- Tape
When you have finished building your cone snail model, describe the cone snail structures you included, as well as the materials you used to make each structure. How do the materials you chose mimic the function of each cone snail structure? Use this Cone Snail Model Evaluation Worksheet to justify your design decisions.
Teachers, you can evaluate student models using this cone snail model evaluation rubric.
Cone Snail Venom Is A Cocktail

Cone snail venom is like a cup of fruit cocktail–it is actually a mix of several different kinds of venom! The venom of each cone snail species contains several different venoms in the form of peptides–small proteins usually between 10-30 amino acids in length. The venom of each cone snail species contains a unique mix of these venom peptides–also called “conotoxins”–that have evolved over many generations to be effective against the prey, predators, and competitors specific to its niche.
An individual peptide is a three-dimensional structure made out of a string of amino acids strung together like Christmas lights. The string of peptides bends and folds itself into a tangled mass with a very specific shape. The snail’s own genetic code determines the order of amino acids in a conotoxin, which influence the ultimate 3D structure of the conotoxin peptide. Because the amino acid sequence ultimately dictates the three-dimensional shape of the resulting peptide, different sequences of amino acids form peptides with different shapes. The unique shapes of cone snail venom peptides, determine how and where that particular venom works within the body of prey.
The more peptides of different shapes there are in a cone snail’s venom cocktail, the more venom components it has at its disposal for poisoning prey.

Most proteins, peptides included, exhibit four styles or “orders” of structure: Primary, secondary, tertiary, and quaternary structures. In the following activity, you will be modeling the primary structure of conotoxins–their amino acid sequences–with beads. You will also model one example of tertiary structure–in this case the covalent bonds between cysteines–called disulfide bridges.
Disulfide bridges are just one of the many amino-acid interactions that give a peptide its 3D shape. In addition, the position and number of these bridges are particularly valuable for predicting how a venom peptide affects the nervous system of prey and can also provide hints about how they might be used for medicinal purposes in humans.

Model The Structure Of Snail Venom Peptides
Materials (Per Group Of 2-4 Students)
- Snail Peptide Information Sheets
- 19 different types of non-magnetic beads, about 10 of each type per student group
You can use letter beads and this amino acids abbreviation chart, or create your own amino acid key by completing this amino acid bead key template. - 1 type of magnetic bead about 30 per student group that will represent cysteine
You can use any type of bead to represent cysteine, but will need wire or adhesive to form “cysteine bonds” between cysteine pairs if the beads are not magnetic.
- ~8-10 inch piece of thin flexible metal wire for threading beads together
- Optional: tape (to hold the beads onto the ends of wire)
- Scissors or wire-cutters to cut excess wire
Procedure
- Choose the snail whose peptides you would like to model with beads, and look through the peptide information sheet that goes with that snail. Each snail peptide sheet has just a sample of the peptides that the species produces.

- Take a minute to note the natural history information of the snail species you’ve selected, as well as the additional information provided about the peptide sequences provided. Can you find any similarities among the peptides just by looking at their primary amino acid structures?
- Choose one peptide from the sheet to recreate with beads (you can do more later). Using either the amino acids abbreviation chart (if using lettered beads) or tape beads of different shapes and colors to the attached amino acid bead key to guide you, thread beads onto your wire to recreate the cone snail peptide amino acid sequence you were given. Whenever you come upon a “C” in the sequence (which represents cysteine), use one of the magnetic beads. It may help to add a bend or fold to the end of the wire to keep your beads from slipping off as you thread them on.
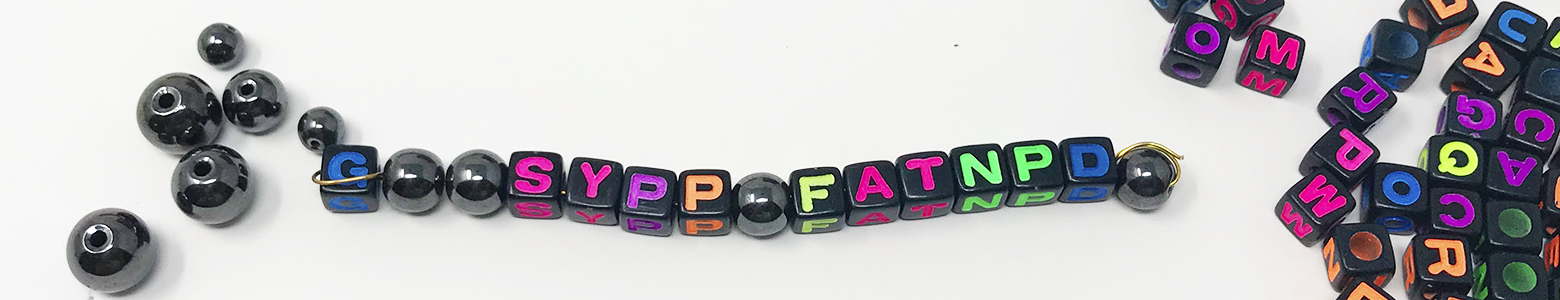
- When you are done, look at the locations of the magnetic cysteine amino acids in your sequence. Can you identify places in the sequence where disulfide bridges could form? (Remember, disulfide bridges are the tertiary structure –the covalent bonds between cysteines). Bend and fold your peptide so that “disulfide bridges” form between your magnetic beads. What 3D peptide conformations can you make in this way?
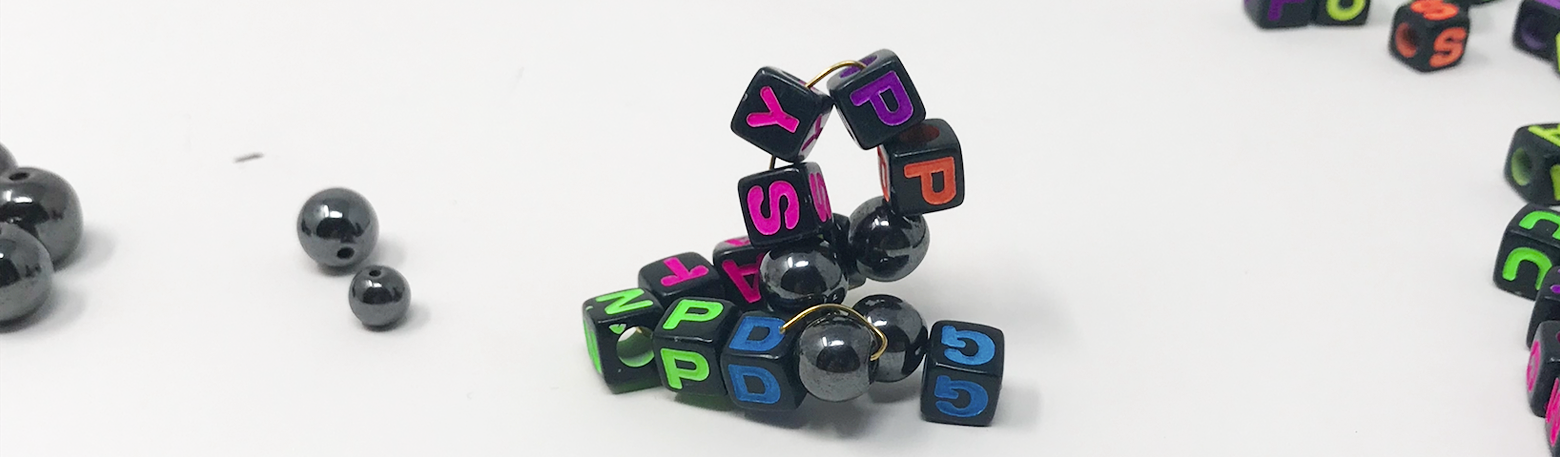
- Next, look at the column labeled “Cysteine Framework”–this column gives a best-guess for which cysteines form disulfide bridges together. A peptide whose cysteine framework reads “1-3, 2-4” most likely has disulfide bridges form between the 1st and 3rd and 2nd and 4th cysteines (reading the sequence left to right). Try folding your cysteine using the recommended cysteine framework. Does it match your original folding prediction? How dramatically does this disulfide bridge configuration change the shape from what you originally predicted?
- Bonus! You may be able to visualize the fully-folded conformation of your peptide by using ConoServer, a website cataloguing cone snail venom peptides. Visit http://www.conoserver.org and type your peptide’s name into the search box, and then select your peptide from the results. If “3D Visualization” is available, click the “Jmol” tab located at the bottom of the screen to see the predicted 3D conformation for your peptide. Using your bead model, try to mimic the folding of your peptide based on the Jmol model that you found, using the magnetic beads to simulate disulfide bonds.
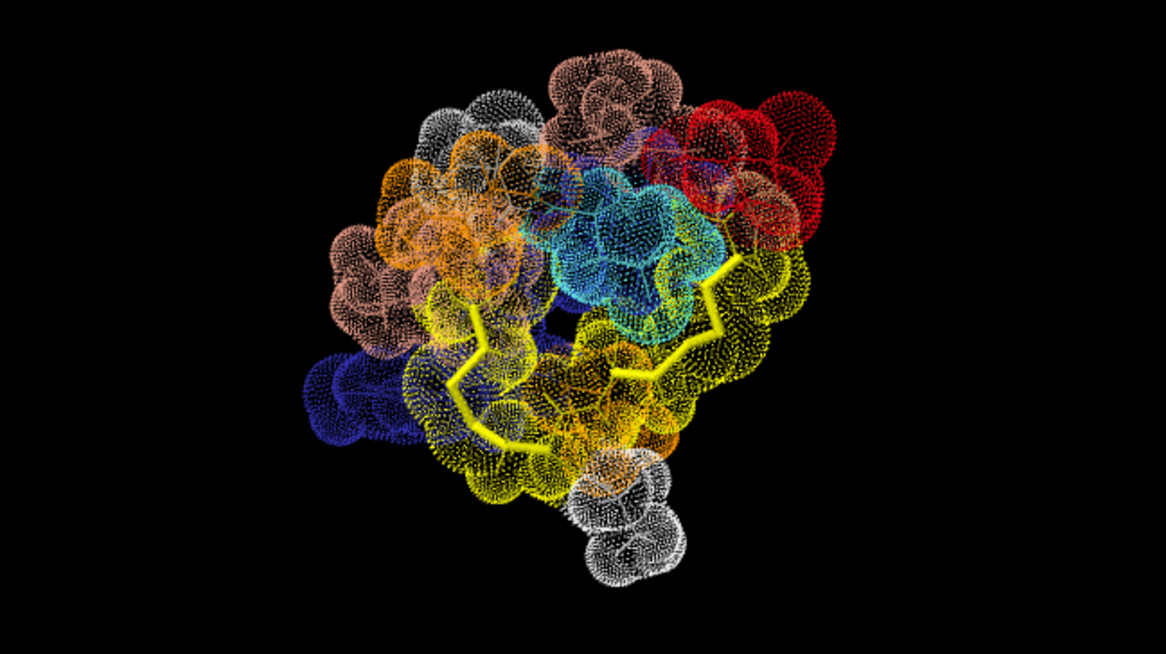
The AuIB conopeptide, from C. aulicus, as visualized with Jmol. Accessed through conoserver.org
Analyze Your Conopeptide Sequences

- Describe how the peptides built by you and your team are visually similar to and different from one another in terms of structure.
- How did connecting the cysteines to each other to form disulfide bridges impact the 3D shape or “conformation” of your peptide?
- What do you think would happen to the shape of the peptides if the disulfide bridges were not formed?
- Look at the other venoms on your Snail Peptide Information Sheet. How are the primary sequences similar and how are they different? Which peptides would you expect to behave similarly in the body based on similarity between structures?
- What could be some advantages to using a cocktail of venom instead of a single toxin to disable prey?
- Find someone with a different cone snail sheet. How do the diets of the different families of cone snails vary? How do their peptide sequences differ from one another? How are they similar?
- Looking at the information sheets provided for the various cone snail peptides, do you see any relationship between the “pharmacological activity” (how it works in the body) and the primary and tertiary structure of the peptide?
- What other patterns do you notice?
- What questions do you have about conotoxins?
Conotoxins As Cures: Mandë Holford The Killer Snail Chemist
By studying the unique behavior, physiology, and venom components of cone snails, researchers like Mandë Holford have been able to find medicinal uses for the toxic peptides in snail venom. Hear Mandë’s story and learn more about how cone snail venom works by watching the video below.
Killer Cone Snails…For Your Health?
Next Generation Science Standards And Notes From The Field
Practice 2: Developing and Using Models
Develop and use a model based on evidence to illustrate the relationships between systems or between components of a system.
Crosscutting Concept: Structure and Function
Investigating or designing new systems or structures requires a detailed examination of the properties of different materials, the structures of different components, and connections of components to reveal its function and/or solve a problem. (HS-LS1-1)
Core Idea LS1:A Structure and Function
Multicellular organisms have a hierarchical structural organization, in which any one system is made up of numerous parts and is itself a component of the next level.
HS-LS1-2. Develop and use a model to illustrate the hierarchical organization of interacting systems that provide specific functions within multicellular organisms.
Additional Curriculum Correlations:
AP Bio (2015): 2.D.1, 2.E.1, 4.A.4, SP1
IB Bio (2016): 2.4, 4.1, B.4
AP Env Sci (2013): II.A
IB Env Systems and Societies (2017): 2.1, 8.2
Common Core (2010): ELA.WHST.6-12.9
Vision and Change (2009): CC2, CC3, DP3
Teachers who field tested this resource had the following advice for teachers adopting this resource:
Bethany from Arizona said: My favorite part was watching them build the models. They went from looking at a basket of miscellaneous items and going, “WHAT???” to seeing cups as shells, clay as a foot, straws as siphons, balloons as organs etc. It was so cool.
Jeff from Illinois said: You can break up the lesson. I did model building one day and protein folding another. I don’t feel kids lost anything by doing it on two separate days.
Credits:
Written by Rebecca Brewer, Biology teacher, Troy School District, Troy, MI
Cone snail illustrations by Jay Rasgorshek
Editing, photos, and data design by Ariel Zych
Editing and digital production by Daniel Peterschimidt
Reviewed by Aileen O’Hearn, Aleeza Oshry, Satoshi Amagai, and Aida Verdes
Educator's Toolbox
Meet the Writers
About Rebecca Brewer
Rebecca Brewer is a high school biology teacher at Troy High School in Michigan. With more than 18 years of experience in the classroom, Rebecca takes a constructivist approach to instruction, which emphasizes student-led learning. She has won several local and national teaching awards and is the director of the Outstanding Biology Teacher Award program for Michigan.
About Ariel Zych
@arieloquentAriel Zych was Science Friday’s director of audience. She is a former teacher and scientist who spends her free time making food, watching arthropods, and being outside.

