Balancing The Good And Bad Of Phosphorus
16:50 minutes
 SciFri Book Club is reading The Devil’s Element by Dan Egan. There’s lots of ways to participate: Read the book, listen to our interview with the author, join our community space, attend an event, or send a message on the SciFri VoxPop app.
SciFri Book Club is reading The Devil’s Element by Dan Egan. There’s lots of ways to participate: Read the book, listen to our interview with the author, join our community space, attend an event, or send a message on the SciFri VoxPop app.
Phosphorus is critical to life as we know it. In fact, every cell in the human body contains this important element.
It’s also a key component in fertilizer. But not all of that fertilizer stays on crops—much of that phosphorus flows into waterways. Therein lies the rub: the runoff fertilizes the plant life growing in the water, creating toxic algal blooms.
To top it all off, the phosphorus reserves in the United States are on track to disappear in just a few decades, according to some estimates.
Ira talks about the past, present, and future of phosphorus with Dan Egan, journalist in residence at the University of Wisconsin-Milwaukee’s School of Freshwater Sciences, and author of the new book, The Devil’s Element: Phosphorus and A World out of Balance.
Want to read The Devil’s Element with us? Join the SciFri Book Club and read along!
Join the SciFri Book Club email newsletter! We send emails a few times per week. If you want to learn more about what we’re reading, you can check out our official Book Club page.
Dan Egan is the author of The Devil’s Element, and Journalist in Residence in the School of Freshwater Sciences at the University of Wisconsin-Milwaukee in Milwaukee, Wisconsin.
IRA FLATOW: This is Science Friday. I’m Ira Flatow. Consider the element phosphorus, number 15 in your periodic table if you’re playing at home. Phosphorus is critical to life as we know it. Every cell in the human body contains phosphorus. It’s a key component in fertilizer, the letter P in your NPK plant food.
But not all of that fertilizer stays on crops. Much of that phosphorus flows into waterways. And there lies the rub. It fertilizes the plant life growing in the water creating toxic algae blooms. And to top it all off, our phosphorus reserves in the US are on track to disappear in just a few decades.
Joining me now to give us a fuller picture about this often overlooked element is my guest Dan Egan, author of the new book and April’s Science Friday Book Club pick, The Devil’s Element– Phosphorus and a World Out of Balance. Dan is a journalist in residence at the University of Wisconsin-Milwaukee School of Freshwater Sciences based in Milwaukee, Wisconsin. Dan, welcome back to Science Friday.
DAN EGAN: Thanks for having me.
IRA FLATOW: Why did you call this book The Devil’s Element?
DAN EGAN: Well, that’s long been the term used for phosphorus. And it’s for a couple of reasons. One is it was the 13th element discovered by an alchemist in Hamburg, Germany back in 1669.
And, two, it’s a pretty dastardly substance. I mean, it is a critical fertilizer, but it’s also been a weapon of war for a very long time. It was known as Willie Pete in Vietnam, white phosphorus. It’s nasty stuff.
IRA FLATOW: OK. Let’s get into this a bit. Let’s start off with phosphorus 101. Phosphorus comes from phosphates, so raw phosphorus does not occur in nature, right?
DAN EGAN: No, it doesn’t. It has to be essentially conjured. And that’s exactly what that alchemist did back in Hamburg, Germany in the 1600s. He was chasing the philosopher’s stone, that magical substance that could turn base metals into gold.
At the time, a lot of people were looking for this mythical substance in a lot of different places. And this particular alchemist, Hennig Brand, he was a urine man. He believed it could be harvested from the human waste stream. And so on his way to trying to make gold, he ended up making elemental phosphorus.
IRA FLATOW: I don’t want to get so much into the great details. But that’s must be a lot of urine to go around collecting.
DAN EGAN: Yeah, yeah. We’re talking vats. I thought of opening this book up with trying to make my own phosphorus because this is a book about phosphorus. And it’s not the sexiest topic. You got to come out of the gate hard.
And so my plan was I have a turkey fryer. I got access to a lot of beer drinking friends and their urine. I had at the time a father-in-law who’s since deceased. But he was a chemical engineer who spent his career working on catalysts for nitrogen production for fertilizer. And he’s this cranky old English guy. And he’s like, we’re not going to get close. But we can have fun trying.
And then I heard from a professor at Johns Hopkins University. And he made it very clear to me that it was a fool’s errand. You weren’t going to get close to making it. And the closer you got, the closer you’re going to get to really hurting yourself because it’s very combustible stuff.
IRA FLATOW: So what was the original phosphorus used for? Was it used for fertilizer?
DAN EGAN: It was mostly just a curiosity. So what condensed from all this urine and from some other hocus-pocus were these waxy little nuggets, maybe the size of chocolate chips. They were cool to the touch, but they cast this little phosphorescent glow. It wasn’t until the 1700s when actual chemists started getting to work, and they realized what it was, and where it could be found, and what it could do to a crop that it became a commodity as a fertilizer.
IRA FLATOW: All right, let’s talk about how we got to use phosphorus as a fertilizer. Tell me how that progressed.
DAN EGAN: Well, ever since agriculture started some 10,000 years ago people have been tinkering with the land, trying to replenish the nutrients lost with the harvest of each crop. And it was a particularly acute problem in Britain because it’s an island nation with limited agriculture lands. And so they were kind of pioneers in all this. And way back, a hundred years ago, they were throwing everything they could think of on crops, blood, fabric, bones, and manure obviously, human and animal manure.
And bones proved to be particularly effective. And so they didn’t really know why at the time. But they knew they wanted bones. And so this propelled the English into some pretty grim places in their never-ending hunt for phosphorus.
Specifically, they went to Waterloo about six years after the battle when some 40,000 people fell in about 10 hours along with a lot of horses. The British came back to plunder that battlefield several years later, stripped it of all of its bones, built some bone crushing mills back in England, and ground them into dust and spread them on crops and made turnips and wheat grow magnificently.
IRA FLATOW: Wow. I mean, you can’t really depend on getting bones to make phosphorus in a large amount for fertilizer, can you?
DAN EGAN: No, no. I mean, the whole story of phosphorus as a fertilizer is this idea that, oh, we’ve found a bountiful source that is never going to play out. And it always plays out. The hunt eventually took them to the West Coast of South America to what’s known as the Guano Islands off of Peru.
And these islands were lousy with phosphorus and, as you mentioned earlier, nitrogen and potassium, the other two critical fertilizing elements. The situation there was it almost never rained in this region. And there were a lot of fish-eating birds. And those birds needed to nest and lay eggs. And they did it on these islands.
And under normal conditions, all the waste they would generate would be washed off the landscape back into the ocean to go into the circle of life. In this case, it just got locked up. And so the British and much of Europe, along with America, began to exploit these islands in the 1840s and ’50s.
And, again, it was thought at the time that we found the mother lode, never going to run out. And in about 40 or 50 years, those islands were pretty much played out. So the hunt went on.
IRA FLATOW: And now most of the phosphorus in the US comes from Florida, right?
DAN EGAN: In the US, yeah. That’s where the primary deposits are. There’s some in Idaho and some in North Carolina. But we’re blowing through them at a pace that– end of the road is coming. We don’t know exactly when. Some say three or four decades, and then we’re going to be on the hunt again.
Most of the phosphorus rock deposits that sustain modern agriculture today are found in Western Sahara and Morocco, 80% of them, as a matter of fact. Think about energy security. Well, there’s workarounds to oil. But there are no workarounds to phosphorus. I mean, every living cell depends on it. And nothing can substitute.
IRA FLATOW: Yeah, because I’ve been reading about news reports talking about is heading towards something they call phosphogeddon.
DAN EGAN: The researchers who coined that phrase, I mean, they’re really trying to get people to think about this because people haven’t been thinking about it for the last century because it seems like we’ve had a bottomless supply. But there’s a bottom there. And also at the same time that we’re blowing through the existing and limited reserves, we’re overusing it in many cases to the point where we’re growing food at the expense of our fresh water because as you mentioned earlier, phosphorus doesn’t lose its fertilizing properties when it washes off a cornfield or a soybean field.
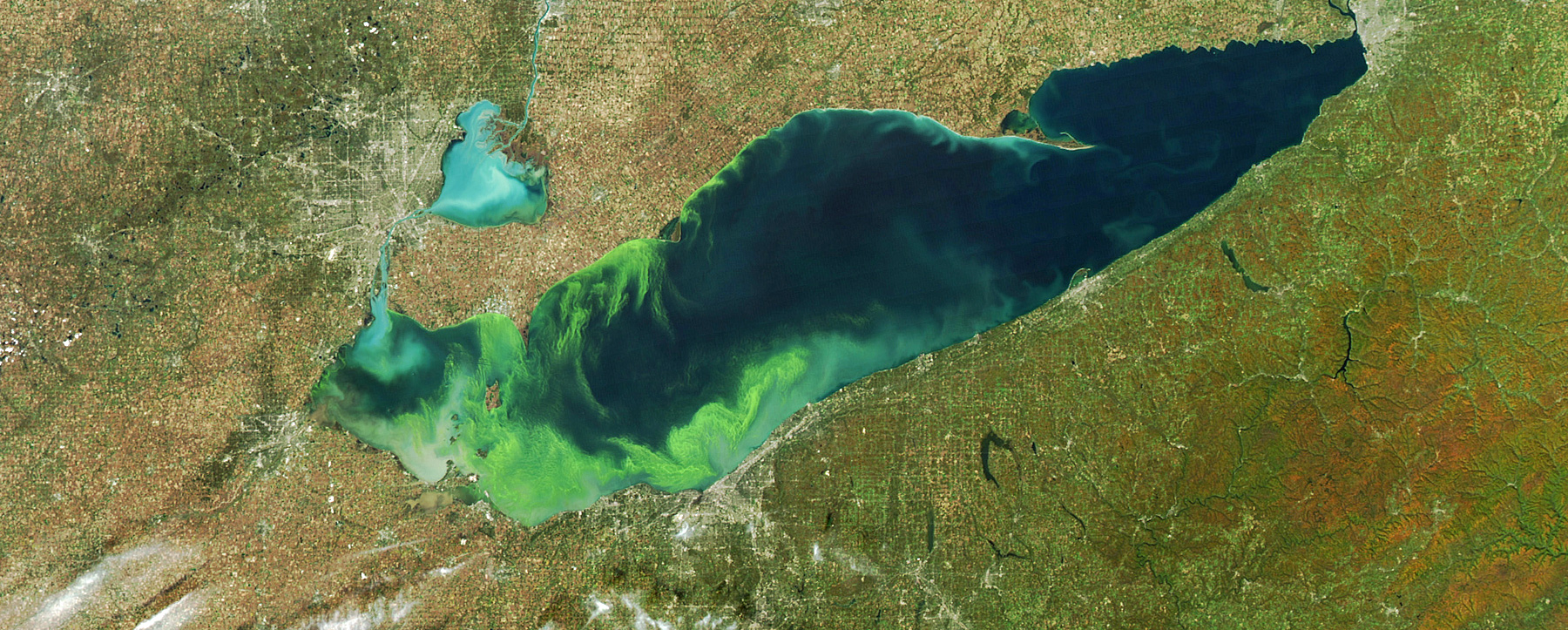
And a lot of it does. And it makes its way into our waters where it is responsible for these horrible algae blooms. These are toxic blooms. It’s blue-green algae, cyanobacteria. They produce toxins that can kill dogs and make people sick. They’ve been suspected in a couple of human deaths over the past couple of decades in the US, including a kid who went swimming in a golf course pond near Madison, Wisconsin.
IRA FLATOW: You– talk about a paradox here because in the sense we’re running out of phosphorus. On the other sense we’re just wasting it as it runs off.
DAN EGAN: Yeah, exactly. And phosphorus really was like the master link in the circle of life. In simple terms, you look at a traditional dairy farm. A cow grazes in a pasture, poops. That poop fertilizes more grass. That cow eats the grass. The cow poops, and on, and on, and on, and on.
And once we figured out that we could squeeze more phosphorus from the earth than it’s naturally willing to cede by mining these specific deposits of rocks, we crack that circle and turned it into a straight line where you use it once, and it just gets flushed away too often. And that’s having dire consequences for our water supplies. So it is a paradox. It’s like we’re running low on it. And we’re also overusing it.
In 2014, Toledo lost its drinking water supply. A half a million people couldn’t safely drink their tap water for several days because of a plume of toxin produced by toxic algae got into their water supply. And you couldn’t boil your way out of this problem because that would only concentrate the toxin. So they’re bringing in pallets– like the National Guard had to come and bring in pallets of baby formula and tanker trucks of water.
And this is a city that’s on the edge of the world’s largest fresh water system. And its residents could not even with treatment safely drink their water for a number of days. They’ve since got the problem fixed or the symptom has been addressed.
But the underlying problem remains. And that is Lake Erie is being overdosed with phosphorous. And that’s producing toxic algae. And that’s threatening humans, and pets, and the environment in general.
IRA FLATOW: So if we are running out of phosphorus, is there a substitute or is the price of phosphorus just going to go through the sky and there are going to be food shortages, things like that?
DAN EGAN: It’s like water. It just keeps circulating and circulating. It may get polluted. It may get locked up in a glacier or something. But the water we have is all the water we’ll ever have, and it doesn’t go away.
And the same thing with phosphorus. But we’re moving it so quickly from the ground, from these stable rock deposits into the living world that at some point we’re going to have to re-stitch this circle of life. We’re going to have to be smarter about reusing the stuff that we put on the fields that don’t get taken up by crops. And manure is a big problem.
A lot of the phosphorus making its way into waters comes from manure. And that is a critical fertilizer. But too often we look at it as just– like these big factory farms, the concentrated animal feeding operations– we look at these lagoons of manure as piles of waste.
And if you think about the British mentality back in the 1800s, they would recognize it for the trove of nutrients that it is. And we’ve got to get back to that point and get smarter about using it. And maybe that’s not just applying raw manure on the ground. But it’s processing it and stripping out the phosphorus, pelletizing it and into a form that’s as pure as anything that’s coming out of a factory. And we can do that. We’re going to have to do it at some point.
IRA FLATOW: This is Science Friday from WNYC Studios. If you’re just joining us, I’m talking with Dan Egan author of the new book The Devil’s Element– Phosphorus and a World Out of Balance. And because of how easily it combusts– I mean, phosphorus burns white hot, doesn’t it? It’s been used as a weapon.
DAN EGAN: Oh, yeah, yeah. And I did encounter a gentleman, and this is not an uncommon phenomenon. He lives north of Hamburg, Germany. And he was ambling along the Baltic Sea looking for just fossilized treasures. A lot of people up on the Baltic coast are always on the hunt for Baltic amber because that used to be a big conifer forest.
And some of the resin from those trees is today amber, and it’s very valued. And nuggets of phosphorus look remarkably like amber. And those nuggets are in the water because– it’s kind of a coincidence. But phosphorous was discovered in Hamburg in 1669. And then Hamburg was burned to the ground by the Allies in 1943 with incendiary bombs.
A lot of them were phosphorous bombs. And those globules– it looks like a firework when a phosphorus bomb explodes. You see these drops of glowing stuff hitting the ground. When it hits a house or a person, it’s going to burn right through them. When it hits water, it stabilizes.
And so there’s all these nuggets that are left over from the bombing of Hamburg. It was seven nights in a row. It was an ungodly amount of stuff dropped on that city. And it’s still there today. And it’s completely stable if it’s in water. But when it’s removed from water and warmed just a tick above room temperature like 80 Fahrenheit, it combusts.
And so I open the book with a guy who thought– I think he thought he had a piece of fossilized oyster shell or something. He just put it in his pocket. And he’s just walking on. He’s by himself, and then his leg just explodes. And he has to go into the sea to put it out. If he leaves– and this is the winter. He’s out there shivering, and shaking, and screaming for help. And they were going to bring in a helicopter, but they thought he’d bring down the helicopter.
So they ended up with an ambulance. And I think they kept the wound wet enough to keep him stable. And they got him to the hospital. And he survived.
But, today, the leg that suffered these burns, he showed it to me. And it looks like tree bark. There’s been so many skin grafts. And this isn’t an unusual occurrence. It’s not like it’s happening every day. But you can Google it, and it happens more than it should, several times a year.
IRA FLATOW: I guess Hamburg is sort of a unifying thread through the history of phosphorus, right?
DAN EGAN: It is. It is. And you were talking earlier about how are we going to find new sources of phosphorus? Well, Germany’s got some really strict rules coming as far as phosphorus discharges from human waste. And they are– I believe it’s in operation now.
They have a state-of-the-art wastewater treatment plant that can recover almost all the phosphorus coming through. So it really does– the story does arc from this alchemist’s chamber in the 1600s to this super state-of-the-art recovery facility on the banks of the Elbe River in 2023.
IRA FLATOW: Interestingly enough, when you write in your book about the supply problem, you’re clear in saying that your book is not intended to be a book of solutions.
DAN EGAN: No, it’s not. And it’s also not intended to disparage the agriculture industry or anything like that. It’s really to connect some dots and to paint a picture that scientists have seen for many years, but the public hasn’t. And that’s just because to date phosphorus has been plentiful and relatively cheap. And people have just kind of learned to live with some of these algae outbreaks.
But according to news reports, I think in 2021 there were like 400 stories of water bodies that had been plagued by this algae. And that was like a 25% increase over the year before. And so it may be a combination of the public just becoming more aware or it may becoming more commonplace. I think it’s probably a bit of both. But we have had the luxury of not having to think about phosphorus as a nutrient and a pollutant for a long time. And we’re losing that now, that luxury.
IRA FLATOW: Dan, great book, very interesting. Thank you for taking time to be with us today.
DAN EGAN: Thanks for having me.
IRA FLATOW: Dan Egan, The Devil’s Element– Phosphorus and a World Out of Balance, that is his new book. Dan is a journalist in residence at the University of Wisconsin-Milwaukee School of Freshwater Sciences based in Milwaukee. If this has whetted your appetite to read the book, well, the Science Friday Book Club members will be reading The Devil’s Element together this April.
And if you’re interested in joining the conversation about our history with phosphorus and we know you’ve been waiting for it, go to sciencefriday.com/element. You can sign up for our newsletter, RSVP for upcoming events, and even enter to win a free book. Once again, that’s sciencefriday.com/element.
Copyright © 2023 Science Friday Initiative. All rights reserved. Science Friday transcripts are produced on a tight deadline by 3Play Media. Fidelity to the original aired/published audio or video file might vary, and text might be updated or amended in the future. For the authoritative record of Science Friday’s programming, please visit the original aired/published recording. For terms of use and more information, visit our policies pages at http://www.sciencefriday.com/about/policies/.
Shoshannah Buxbaum is a producer for Science Friday. She’s particularly drawn to stories about health, psychology, and the environment. She’s a proud New Jersey native and will happily share her opinions on why the state is deserving of a little more love.
Diana Plasker was the Senior Manager of Experiences at Science Friday, where she created live events, programs and partnerships to delight and engage audiences in the world of science.
Ira Flatow is the founder and host of Science Friday. His green thumb has revived many an office plant at death’s door.