16:51
Vines Choking Out Trees in the Tropics
Increased forest fragmentation and a boost in carbon dioxide may contribute to the vines’ success.
A Double Take on the Northern Lights
A method using consumer grade photography gear offers a fresh look at the aurora borealis.
21:34
‘Countdown’ Explores the Effects of Our Overpopulated Planet
How can we sustain and balance the exploding global population?
23:32
Saving Wild Places in the ‘Anthropocene’
Do we need to rethink how we protect nature in the age of climate change?
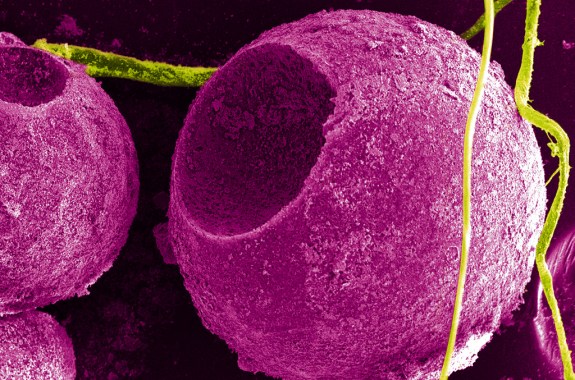
Picture of the Week: Quantum Dot Sphere
This olive-like structure is composed of tiny luminescing spheres that could be used in cellular imaging.
Chasing Time Machines: A Photographer Turns High Energy Physics Into Art
Stanley Greenberg takes pictures of giant structures used to study tiny particles.
9:33
Why Climate Change Ups the Odds of Fires, Floods
Climatologist Jennifer Francis talks about how climate change and severe weather may be linked.
29:07
Can Mass Transit Solve City Sprawl?
L.A. commuters spend 60 hours a year stuck in traffic. But is mass transit a viable alternative?
13:06
Birding the Fall Migration
Birding tips on how to get the most out of the fall bird migration.
Here’s What a Fire Whirl Looks Like
Firefighter Greg Sanders encountered a flaming, cobra-like swirl while doing reconnaissance work in Virginia.