Media Guide: HIV Prevention With PrEP
Use this classroom resource to have your students learn about PrEP, an HIV prevention treatment that is the subject of a recent study in the Netherlands. Discuss implications of PrEP on the spread of HIV with this audio segment from Science Friday.
Make A Mathy Valentine
Spice up your valentines this year by using a little geometry to create consistent hearts and captivating patterns.
Teachers Bring Fracking to School
Looking to inject a little fracking into your curriculum? Check out these resources developed by Colorado educators in the AirWaterGas Teachers-in-Residence program.
Make A Home For Microbes To Understand Their Behavior
Build a home for microbes as you create a Winogradsky column, a simple ecosystem for bacteria and other microorganisms.
High Pressure In The Deep Ocean
Pressure is a huge challenge for deep ocean explorers. Learn how pressure changes with depth and explore its effects on compressible solids in this series of experiments, demonstrations, and real-life data collected aboard the E/V Nautilus.
The Tragic Mystery Of The Mushy Apple
In this experiment, you’ll explore the influence of apple cell structure on the crunchiness and juiciness of an apple by measuring apple tissue tensile strength.
The Science Club #MessageChallenge
Invent a device or system that can send or carry a message from one place to another.
Smelly Bats
A fun game for Halloween that demonstrates diffusion and the properties of stretchy polymers using rubber gloves and flavor extracts.
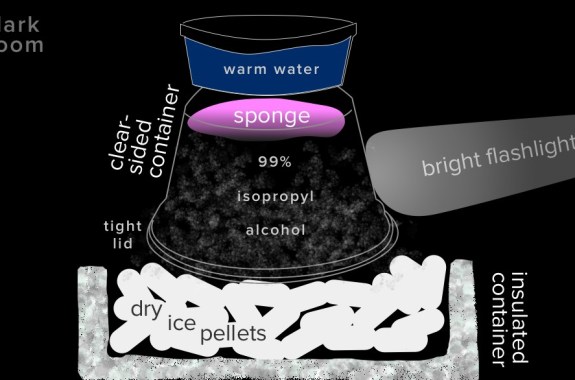
Build A Cloud Chamber
Observe the radioactive particles all around you by building a cloud chamber using a clear container, dry ice, and a little rubbing alcohol.
Go Out And Observe the Moon!
Observe the moon each day for one month, map the moon’s features, simulate lunar impacts, and explore lunar science in this resource from NASA’s International Observe the Moon Night (InOMN), an annual worldwide celebration of lunar science and exploration.